Activation of the nonreceptor protein tyrosine kinase Ack by multiple extracellular stimuli
- PMID: 16777958
- PMCID: PMC1502533
- DOI: 10.1073/pnas.0603714103
Activation of the nonreceptor protein tyrosine kinase Ack by multiple extracellular stimuli
Abstract
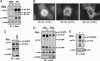
Ack/Ack1 is a nonreceptor protein tyrosine kinase that comprises a tyrosine kinase core, an SH3 domain, a Cdc42-binding region, a Ralt homology region, and a proline-rich region. Here we describe a detailed characterization of the Ack protein as well as the chromosomal localization of human Ack (chromosome 3q29) and the primary structure of murine Ack. We demonstrate that Ack is ubiquitously expressed, with highest expression seen in thymus, spleen, and brain. Activation of integrins by cell adhesion on fibronectin leads to strong tyrosine phosphorylation and activation of Ack. Upon cell stimulation with EGF or PDGF, Ack is tyrosine-phosphorylated and recruited to activated EGF or PDGF receptors, respectively. A pool of endogenous Ack molecules is constitutively tyrosine-phosphorylated, even in starved cells. Moreover, tyrosine-phosphorylated Ack forms a stable complex with the adapter protein Nck via its SH2 domain. Finally, we have characterized a membrane-targeting sterile alpha motif-like domain in the amino terminus of Ack. Using several Ack mutants, we show that the amino-terminal and CRIB domains are necessary for Ack autophosphorylation, whereas the SH3 domain appears to have an autoinhibitory role. These experiments suggest a functional role for Ack as an early transducer of multiple extracellular stimuli.
Conflict of interest statement
Conflict of interest statement: No conflicts declared.
Figures
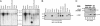
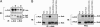


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

