Proteomic identification of desmoglein 2 and activated leukocyte cell adhesion molecule as substrates of ADAM17 and ADAM10 by difference gel electrophoresis
- PMID: 16782893
- PMCID: PMC1489169
- DOI: 10.1128/MCB.02380-05
Proteomic identification of desmoglein 2 and activated leukocyte cell adhesion molecule as substrates of ADAM17 and ADAM10 by difference gel electrophoresis
Abstract
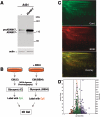
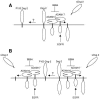
In contrast with the early view of metalloproteases as simple extracellular matrix-degrading entities, recent findings show that they are highly specific modulators of different signaling pathways involved, positively or negatively, in tumor development. Thus, before considering a given metalloprotease a therapeutic target, it seems advisable to characterize its function by identifying its repertoire of substrates. Here, we present a proteomic approach to identify ADAM17 substrates by difference gel electrophoresis. We found that the shedding of the extracellular domain of the transferrin receptor and those of two cell-cell adhesion molecules, activated leukocyte cell adhesion molecule (ALCAM) and desmoglein 2 (Dsg-2), is increased in cells overexpressing ADAM17. Genetic evidence shows that while ADAM17 is responsible for the shedding of ALCAM, both ADAM17 and ADAM10 can act on Dsg-2. Activation of the epidermal growth factor receptor leads to the upregulation of the shedding of Dsg-2 and to the concomitant upregulation of ADAM17, but not ADAM10, supporting the ability of overexpressed ADAM17 to shed Dsg-2. These results unveil a role of ADAM10 and ADAM17 in the shedding of cell-cell adhesion molecules. Since loss of cell adhesion is an early event in tumor development, these results suggest that ADAM17 is a useful target in anticancer therapy.
Figures



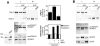
References
-
- Annaert, W., and B. De Strooper. 2002. A cell biological perspective on Alzheimer's disease. Annu. Rev. Cell Dev. Biol. 18:25-51. - PubMed
-
- Arribas, J., and A. Borroto. 2002. Protein ectodomain shedding. Chem. Rev. 102:4627-4638. - PubMed
-
- Baker, A. H., D. R. Edwards, and G. Murphy. 2002. Metalloproteinase inhibitors: biological actions and therapeutic opportunities. J. Cell Sci. 115:3719-3727. - PubMed
-
- Balbin, M., A. Fueyo, A. M. Tester, A. M. Pendas, A. S. Pitiot, A. Astudillo, C. M. Overall, S. D. Shapiro, and C. Lopez-Otin. 2003. Loss of collagenase-2 confers increased skin tumor susceptibility to male mice. Nat. Genet. 35:252-257. - PubMed
-
- Baselga, J., and C. L. Arteaga. 2005. Critical update and emerging trends in epidermal growth factor receptor targeting in cancer J. Clin. Oncol. 23:2445-2459. (Erratum, 23:6281.) - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
