Adenosine A(2A) receptors play a role in the pathogenesis of hepatic cirrhosis
- PMID: 16783407
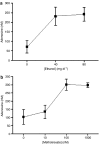
- PMCID: PMC1752015
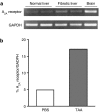
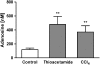
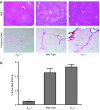
- DOI: 10.1038/sj.bjp.0706812
Adenosine A(2A) receptors play a role in the pathogenesis of hepatic cirrhosis
Abstract
1. Adenosine is a potent endogenous regulator of inflammation and tissue repair. Adenosine, which is released from injured and hypoxic tissue or in response to toxins and medications, may induce pulmonary fibrosis in mice, presumably via interaction with a specific adenosine receptor. We therefore determined whether adenosine and its receptors contribute to the pathogenesis of hepatic fibrosis. 2. As in other tissues and cell types, adenosine is released in vitro in response to the fibrogenic stimuli ethanol (40 mg dl(-1)) and methotrexate (100 nM). 3. Adenosine A(2A) receptors are expressed on rat and human hepatic stellate cell lines and adenosine A(2A) receptor occupancy promotes collagen production by these cells. Liver sections from mice treated with the hepatotoxins carbon tetrachloride (CCl(4)) (0.05 ml in oil, 50 : 50 v : v, subcutaneously) and thioacetamide (100 mg kg(-1) in PBS, intraperitoneally) released more adenosine than those from untreated mice when cultured ex vivo. 4. Adenosine A(2A) receptor-deficient, but not wild-type or A(3) receptor-deficient, mice are protected from development of hepatic fibrosis following CCl(4) or thioacetamide exposure. 5. Similarly, caffeine (50 mg kg(-1) day(-1), po), a nonselective adenosine receptor antagonist, and ZM241385 (25 mg kg(-1) bid), a more selective antagonist of the adenosine A(2A) receptor, diminished hepatic fibrosis in wild-type mice exposed to either CCl(4) or thioacetamide. 6. These results demonstrate that hepatic adenosine A(2A) receptors play an active role in the pathogenesis of hepatic fibrosis, and suggest a novel therapeutic target in the treatment and prevention of hepatic cirrhosis.
Figures



References
-
- AGUADO J.M., PONS F., CASAFONT F., SAN MIGUEL G., VALLE R. Tuberculous peritonitis: a study comparing cirrhotic and noncirrhotic patients. J. Clin. Gastroenterol. 1990;12:550–554. - PubMed
-
- ANDERSSON S.E., JOHANSSON L.H., LEXMULLER K., EKSTROM G.M. Anti-arthritic effect of methotrexate: is it really mediated by adenosine. Eur. J. Pharm. Sci. 2000;9:333–343. - PubMed
-
- BSHESH K., ZHAO B., SPIGHT D., BIAGGIONI I., FEOKISTOV I., DENENBERG A., WONG H.R., SHANLEY T.P. The A2A receptor mediates an endogenous regulatory pathway of cytokine expression in THP-1 cells. J. Leukoc. Biol. 2002;72:1027–1036. - PubMed
-
- BURGESS L.J., SWANEPOEL C.G., TALJAARD J.J. The use of adenosine deaminase as a diagnostic tool for peritoneal tuberculosis. Tuberculosis (Edinb) 2001;81:243–248. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

