Zymosan-induced glycerylprostaglandin and prostaglandin synthesis in resident peritoneal macrophages: roles of cyclo-oxygenase-1 and -2
- PMID: 16787386
- PMCID: PMC1570173
- DOI: 10.1042/BJ20060615
Zymosan-induced glycerylprostaglandin and prostaglandin synthesis in resident peritoneal macrophages: roles of cyclo-oxygenase-1 and -2
Abstract
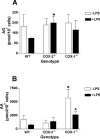
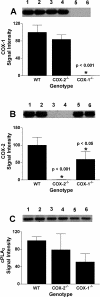
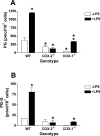
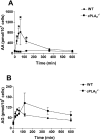
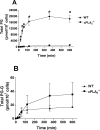
COX [cyclo-oxygenase; PG (prostaglandin) G/H synthase] oxygenates AA (arachidonic acid) and 2-AG (2-arachidonylglycerol) to endoperoxides that are converted into PGs and PG-Gs (glycerylprostaglandins) respectively. In vitro, 2-AG is a selective substrate for COX-2, but in zymosan-stimulated peritoneal macrophages, PG-G synthesis is not sensitive to selective COX-2 inhibition. This suggests that COX-1 oxygenates 2-AG, so studies were carried out to identify enzymes involved in zymosan-dependent PG-G and PG synthesis. When macrophages from COX-1-/- or COX-2-/- mice were treated with zymosan, 20-25% and 10-15% of the PG and PG-G synthesis observed in wild-type cells respectively was COX-2 dependent. When exogenous AA and 2-AG were supplied to COX-2-/- macrophages, PG and PG-G synthesis was reduced as compared with wild-type cells. In contrast, when exogenous substrates were provided to COX-1-/- macrophages, PG-G but not PG synthesis was reduced. Product synthesis also was evaluated in macrophages from cPLA(2alpha) (cytosolic phospholipase A2alpha)-/- mice, in which zymosan-induced PG synthesis was markedly reduced, and PG-G synthesis was increased approx. 2-fold. These studies confirm that peritoneal macrophages synthesize PG-Gs in response to zymosan, but that this process is primarily COX-1-dependent, as is the synthesis of PGs. They also indicate that the 2-AG and AA used for PG-G and PG synthesis respectively are derived from independent pathways.
Figures

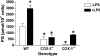
References
-
- Nugteren D. H., Hazelhof E. Isolation and properties of intermediates in prostaglandin biosynthesis. Biochim. Biophys. Acta. 1973;326:448–461. - PubMed
-
- Rouzer C. A., Marnett L. J. Mechanism of free radical oxygenation of polyunsaturated fatty acids by cyclooxygenases. Chem. Rev. 2003;103:2239–2304. - PubMed
-
- Smith W. L., DeWitt D. L., Garavito R. M. Cyclooxygenases: structural, cellular, and molecular biology. Annu. Rev. Biochem. 2000;69:145–182. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- P30 HD033994/HD/NICHD NIH HHS/United States
- DA06668/DA/NIDA NIH HHS/United States
- R37 HD012304/HD/NICHD NIH HHS/United States
- P01-CA-77839/CA/NCI NIH HHS/United States
- GM15431/GM/NIGMS NIH HHS/United States
- HD12304/HD/NICHD NIH HHS/United States
- P01 CA077839/CA/NCI NIH HHS/United States
- P01 GM015431/GM/NIGMS NIH HHS/United States
- P50 GM015431/GM/NIGMS NIH HHS/United States
- R01 DA006668/DA/NIDA NIH HHS/United States
- R37 DA006668/DA/NIDA NIH HHS/United States
- U54 HD033994/HD/NICHD NIH HHS/United States
- HD33994/HD/NICHD NIH HHS/United States
LinkOut - more resources
Full Text Sources
Research Materials

