Bacillus anthracis phospholipases C facilitate macrophage-associated growth and contribute to virulence in a murine model of inhalation anthrax
- PMID: 16790747
- PMCID: PMC1489738
- DOI: 10.1128/IAI.00307-06
Bacillus anthracis phospholipases C facilitate macrophage-associated growth and contribute to virulence in a murine model of inhalation anthrax
Abstract
Several models of anthrax pathogenesis suggest that early in the infectious process Bacillus anthracis endospores germinate and outgrow into vegetative bacilli within phagocytes before being released into the blood. Here, we define the respective contributions of three phospholipases C (PLCs) to the pathogenesis of B. anthracis. Genetic deletions of the PLCs were made in the Sterne 7702 background, resulting in the respective loss of their activities. The PLCs were redundant both in tissue culture and in murine models of anthrax. Deletion of all three PLC genes was required for attenuation of virulence in mice after intratracheal inoculation. This attenuation may be attributed to the inability of the PLC-null strain to grow in association with the macrophage. Complementation of these defects in both models of anthrax was achieved by expression of the PLC genes in trans. The functional redundancy between PLCs in the virulence of B. anthracis implies that their activities are important for anthrax pathogenesis.
Figures

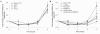
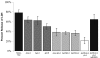

References
-
- Burkholder, P. R., and N. H. Giles. 1947. Induced biochemical mutations in Bacillus subtilis. Am. J. Bot. 34:345-348. - PubMed
-
- Cendrowski, S., W. MacArthur, and P. Hanna. 2004. Bacillus anthracis requires siderophore biosynthesis for growth in macrophages and mouse virulence. Mol. Microbiol. 51:407-417. - PubMed
-
- Dixon, T. C., A. A. Fadl, T. M. Koehler, J. A. Swanson, and P. C. Hanna. 2000. Early Bacillus anthracis-macrophage interactions: intracellular survival and escape. Cell. Microbiol. 2:453-463. - PubMed
-
- Dixon, T. C., M. Meselson, J. Guillemin, and P. C. Hanna. 1999. Anthrax. N. Engl. J. Med. 341:815-826. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

