Invasion of epithelial cells and proteolysis of cellular focal adhesion components by distinct types of Porphyromonas gingivalis fimbriae
- PMID: 16790749
- PMCID: PMC1489697
- DOI: 10.1128/IAI.01902-05
Invasion of epithelial cells and proteolysis of cellular focal adhesion components by distinct types of Porphyromonas gingivalis fimbriae
Abstract
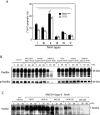
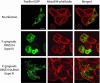
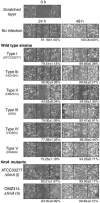
Porphyromonas gingivalis fimbriae are classified into six types (types I to V and Ib) based on the fimA genes encoding FimA (a subunit of fimbriae), and they play a critical role in bacterial interactions with host tissues. In this study, we compared the efficiencies of P. gingivalis strains with distinct types of fimbriae for invasion of epithelial cells and for degradation of cellular focal adhesion components, paxillin, and focal adhesion kinase (FAK). Six representative strains with the different types of fimbriae were tested, and P. gingivalis with type II fimbriae (type II P. gingivalis) adhered to and invaded epithelial cells at significantly greater levels than the other strains. There were negligible differences in gingipain activities among the six strains; however, type II P. gingivalis apparently degraded intracellular paxillin in association with a loss of phosphorylation 30 min after infection. Degradation was blocked with cytochalasin D or in mutants with fimA disrupted. Paxillin was degraded by the mutant with Lys-gingipain disrupted, and this degradation was prevented by inhibition of Arg-gingipain activity by Nalpha-p-tosyl-l-lysine chloromethyl ketone. FAK was also degraded by type II P. gingivalis. Cellular focal adhesions with green fluorescent protein-paxillin macroaggregates were clearly destroyed, and this was associated with cellular morphological changes and microtubule disassembly. In an in vitro wound closure assay, type II P. gingivalis significantly inhibited cellular migration and proliferation compared to the cellular migration and proliferation observed with the other types. These results suggest that type II P. gingivalis efficiently invades epithelial cells and degrades focal adhesion components with Arg-gingipain, which results in cellular impairment during wound healing and periodontal tissue regeneration.
Figures



References
-
- Amano, A., I. Nakagawa, N. Okahashi, and N. Hamada. 2004. Variations of Porphyromonas gingivalis fimbriae in relation to microbial pathogenesis. J. Periodontal Res. 39:136-142. - PubMed
-
- Amano, A., M. Kuboniwa, I. Nakagawa, S. Akiyama, S., I. Morisaki, and S. Hamada. 2000. Prevalence of specific genotypes of Porphyromonas gingivalis fimA and periodontal health status. J. Dent. Res. 79:1664-1668. - PubMed
-
- Belton, C. M., K. T. Izutsu, P. C. Goodwin, Y. Park, and R. J. Lamont. 1999. Fluorescence image analysis of the association between Porphyromonas gingivalis and gingival epithelial cells. Cell. Microbiol. 1:215-223. - PubMed
-
- Chen, Z., C. A. Casiano, and H. M. Fletcher. 2001. Protease-active extracellular protein preparations from Porphyromonas gingivalis W83 induce N-cadherin proteolysis, loss of cell adhesion, and apoptosis in human epithelial cells. J. Periodontol. 72:641-650. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

