Insulin-like growth factor-1 (IGF-1) induces the activation/phosphorylation of Akt kinase and cAMP response element-binding protein (CREB) by activating different signaling pathways in PC12 cells
- PMID: 16792806
- PMCID: PMC1534052
- DOI: 10.1186/1471-2202-7-51
Insulin-like growth factor-1 (IGF-1) induces the activation/phosphorylation of Akt kinase and cAMP response element-binding protein (CREB) by activating different signaling pathways in PC12 cells
Abstract
Background: Insulin-like growth factor-1 (IGF-1) is a polypeptide growth factor with a variety of functions in both neuronal and non-neuronal cells. IGF-1 plays anti-apoptotic and other functions by activating multiple signaling pathways including Akt kinase, a serine/threonine kinase essential for cell survival. The nuclear transcription factor cAMP response element-binding protein (CREB) may also be involved although relationships between these two proteins in IGF-1 receptor signaling and protection is not clear, especially in neuronal cells.
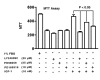
Results: IGF-1, in a concentration- and time-dependent manner, induces the activation/phosphorylation of Akt and CREB in PC12 cells by activating different signaling pathways. IGF-1 induced a sustained phosphorylation of Akt while only a transient one was seen for CREB. The phosphorylation of Akt is mediated by the PI3 kinase pathway while that of CREB is dependent on the activation of both MAPK kinase and p38 MAPK. Moreover, the stimulation of PKC attenuated the phosphorylation of Akt induced by IGF-1 while enhancing that of CREB. Survival assays with various kinase inhibitors suggested that the activation/phosphorylation of both Akt and CREB contributes to IGF-1 mediated cell survival in PC12 cells.
Conclusion: These data suggest that IGF-1 induced the activation of Akt and CREB using distinct pathways in PC12 cells.
Figures








References
-
- Zheng WH, Kar S, Quirion R. Insulin-like growth factor-1-induced phosphorylation of transcription factor FKHRL1 is mediated by phosphatidylinositol 3-kinase/Akt kinase and role of this pathway in insulin-like growth factor-1-induced survival of cultured hippocampal neurons. Mol Pharmacol. 2002;62:225–233. doi: 10.1124/mol.62.2.225. - DOI - PubMed
-
- Sasaoka T, Rose DW, Jhun BH, Saltiel AR, Draznin B, Olefsky JM. Evidence for a functional role of Shc proteins in mitogenic signaling induced by insulin, insulin-like growth factor-1, and epidermal growth factor. J Biol Chem. 1994;269:13689–13694. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

