Photodynamic therapy and anti-tumour immunity
- PMID: 16794636
- PMCID: PMC2933780
- DOI: 10.1038/nrc1894
Photodynamic therapy and anti-tumour immunity
Abstract
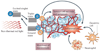
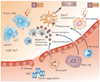
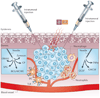
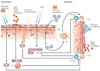
Photodynamic therapy (PDT) uses non-toxic photosensitizers and harmless visible light in combination with oxygen to produce cytotoxic reactive oxygen species that kill malignant cells by apoptosis and/or necrosis, shut down the tumour microvasculature and stimulate the host immune system. In contrast to surgery, radiotherapy and chemotherapy that are mostly immunosuppressive, PDT causes acute inflammation, expression of heat-shock proteins, invasion and infiltration of the tumour by leukocytes, and might increase the presentation of tumour-derived antigens to T cells.
Figures

References
-
- Moan J, Peng Q. An outline of the hundred-year history of PDT. Anticancer Res. 2003;23:3591–3600. - PubMed
-
-
Dolmans DE, Fukumura D, Jain RK. Photodynamic therapy for cancer. Nature Rev. Cancer. 2003;3:380–387. A recent review on the theory and applications of PDT in cancer treatment.
-
-
- Detty MR, Gibson SL, Wagner SJ. Current clinical and preclinical photosensitizers for use in photodynamic therapy. J. Med. Chem. 2004;47:3897–3915. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

