An update on the first decade of the European centralized procedure: how many innovative drugs?
- PMID: 16796703
- PMCID: PMC1885166
- DOI: 10.1111/j.1365-2125.2006.02700.x
An update on the first decade of the European centralized procedure: how many innovative drugs?
Abstract
Aims: In a previous paper, we proposed an algorithm to assess the degree of therapeutic innovation of the agents approved by the European centralized procedure, which must be followed by biotechnological products and is optional for drugs claimed as innovative. A low overall degree of therapeutic innovation (about 30%) was found. This figure may be an underestimate of the actual level of innovation, because common biotechnological products, such as recombinant human insulins, must follow this procedure. To test the hypothesis that therapeutic innovation prevails among nonbiotechnological products, we evaluated separately the degree of therapeutic innovation of biotechnological vs. nonbiotechnological agents in the first decade of European Medicines Agency activity, also studying a possible time trend.
Methods: We assessed, for each drug: (i) the seriousness of the target disease, (ii) the availability of previous treatments, and (iii) the extent of therapeutic effect according to the previously proposed algorithm.
Results: Our analysis considered 251 medicinal products corresponding to 198 active substances, classified according to four main areas as therapeutic agents (88.9%), diagnostics (5.5%), vaccines (5.1%) and life-style drugs (0.5%). Among all therapeutic agents, 49 out of 176 agents (28%) were classified as having an important degree of therapeutic innovation. Fifteen out of 60 biotechnological therapeutic agents were considered important therapeutic innovations (25%), whereas this figure was 29% for nonbiotechnological agents.
Conclusions: Among active substances claimed as innovative by the manufacturers, only a minority deserve this definition according to our algorithm.
Figures
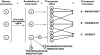

 ), nonbiotechnological (
), nonbiotechnological ( )
)
 ), moderate (
), moderate ( ), modest (
), modest ( ), pharmacological (
), pharmacological ( ), tecnological (▪)
), tecnological (▪)Comment in
-
On criteria to evaluate the therapeutic innovation of drugs.Br J Clin Pharmacol. 2021 May;87(5):2403-2404. doi: 10.1111/bcp.14650. Epub 2020 Nov 20. Br J Clin Pharmacol. 2021. PMID: 33219565 No abstract available.
References
-
- The Council of the European Communities. Council Regulation (EEC) no2309/93 of 22 July 1993 laying down Community procedures for the authorization and supervision of medicinal products for human and veterinary use and establishing a European Agency for the Evaluation of Medicinal Products. Regulation (EEC) No. 2309/93.
-
- Garattini S, Bertelé V. Efficacy, safety and cost of new cardiovascular drugs: a survey. Eur J Clin Pharmacol. 2003;59:701–6. - PubMed
-
- Garattini S, Bertelé V. Efficacy, safety and cost of new drugs acting on the central nervous system. Eur J Clin Pharmacol. 2003;59:79–84. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

