Palytoxin-induced effects on partial reactions of the Na,K-ATPase
- PMID: 16801384
- PMCID: PMC2151552
- DOI: 10.1085/jgp.200609505
Palytoxin-induced effects on partial reactions of the Na,K-ATPase
Abstract
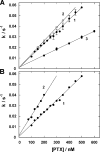
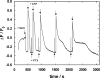
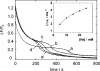
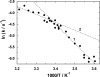
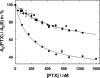
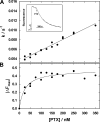
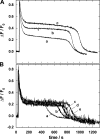
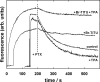
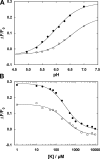
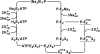
The interaction of palytoxin with the Na,K-ATPase was studied by the electrochromic styryl dye RH421, which monitors the amount of ions in the membrane domain of the pump. The toxin affected the pump function in the state P-E2, independently of the type of phosphorylation (ATP or inorganic phosphate). The palytoxin-induced modification of the protein consisted of two steps: toxin binding and a subsequent conformational change into a transmembrane ion channel. At 20 degrees C, the rate-limiting reaction had a forward rate constant of 10(5) M(-1)s(-1) and a backward rate constant of about 10(-3) s(-1). In the palytoxin-modified state, the binding affinity for Na+ and H+ was increased and reached values between those obtained in the E1 and P-E2 conformation under physiological conditions. Even under saturating palytoxin concentrations, the ATPase activity was not completely inhibited. In the Na/K mode, approximately 50% of the enzyme remained active in the average, and in the Na-only mode 25%. The experimental findings indicate that an additional exit from the inhibited state exists. An obvious reaction pathway is a slow dephosphorylation of the palytoxin-inhibited state with a time constant of approximately 100 s. Analysis of the effect of blockers of the extracellular and cytoplasmic access channels, TPA+ and Br2-Titu3+, respectively, showed that both access channels are part of the ion pathway in the palytoxin-modified protein. All experiments can be explained by an extension of the Post-Albers cycle, in which three additional states were added that branch off in the P-E2 state and lead to states in which the open-channel conformation is introduced and returns into the pump cycle in the occluded E2 state. The previously suggested molecular model for the channel state of the Na,K-ATPase as a conformation in which both gates between binding sites and aqueous phases are simultaneously in their open state is supported by this study.
Figures

Similar articles
-
Stopped-flow kinetic investigations of conformational changes of pig kidney Na+,K+-ATPase.Biochemistry. 1997 Oct 28;36(43):13406-20. doi: 10.1021/bi970598w. Biochemistry. 1997. PMID: 9341234
-
Kinetics of the phosphorylation of Na,K-ATPase by inorganic phosphate detected by a fluorescence method.Biochemistry. 1996 Aug 20;35(33):10922-30. doi: 10.1021/bi960238t. Biochemistry. 1996. PMID: 8718885
-
Palytoxin and the sodium/potassium pump--phosphorylation and potassium interaction.Phys Biol. 2009 May 21;6(3):036010. doi: 10.1088/1478-3975/6/3/036010. Phys Biol. 2009. PMID: 19461129
-
Partial reactions of the Na,K-ATPase: kinetic analysis and transport properties.Acta Physiol Scand Suppl. 1998 Aug;643:235-45. Acta Physiol Scand Suppl. 1998. PMID: 9789566 Review.
-
The partial reactions of the Na(+)- and Na(+) + K(+)-activated adenosine triphosphatases.Soc Gen Physiol Ser. 1991;46:227-47. Soc Gen Physiol Ser. 1991. PMID: 1653982 Review.
Cited by
-
Visualizing the mapped ion pathway through the Na,K-ATPase pump.Channels (Austin). 2009 Nov;3(6):383-6. doi: 10.4161/chan.3.6.9775. Epub 2009 Nov 10. Channels (Austin). 2009. PMID: 19806033 Free PMC article. Review.
-
Kinetic contribution to extracellular Na+/K+ selectivity in the Na+/K+ pump.FEBS Open Bio. 2018 Apr 16;8(5):854-859. doi: 10.1002/2211-5463.12418. eCollection 2018 May. FEBS Open Bio. 2018. PMID: 29744299 Free PMC article.
-
The structure of the neurotoxin palytoxin determined by MicroED.bioRxiv [Preprint]. 2023 Jun 1:2023.03.31.535166. doi: 10.1101/2023.03.31.535166. bioRxiv. 2023. PMID: 37034718 Free PMC article. Preprint.
-
Sodium flux ratio in Na/K pump-channels opened by palytoxin.J Gen Physiol. 2007 Jul;130(1):41-54. doi: 10.1085/jgp.200709770. Epub 2007 Jun 11. J Gen Physiol. 2007. PMID: 17562821 Free PMC article.
-
Maitotoxin converts the plasmalemmal Ca(2+) pump into a Ca(2+)-permeable nonselective cation channel.Am J Physiol Cell Physiol. 2009 Dec;297(6):C1533-43. doi: 10.1152/ajpcell.00252.2009. Epub 2009 Sep 30. Am J Physiol Cell Physiol. 2009. PMID: 19794142 Free PMC article.
References
-
- Apell, H.J. 2004. How do P-type ATPases transport ions? Bioelectrochemistry. 63:149–156. - PubMed
-
- Apell, H.-J. 2003. Toward an understanding of ion transport through the Na,K-ATPase. Ann. N. Y. Acad. Sci. 986:133–140. - PubMed
-
- Apell, H.-J., and A. Diller. 2002. Do H+ ions obscure electrogenic Na+ and K+ binding in the E1 state of the Na,K-ATPase? FEBS Lett. 532:198–202. - PubMed
-
- Apell, H.-J., M. Roudna, J.E. Corrie, and D.R. Trentham. 1996. Kinetics of the phosphorylation of Na,K-ATPase by inorganic phosphate detected by a fluorescence method. Biochemistry. 35:10922–10930. - PubMed
-
- Artigas, P., and D.C. Gadsby. 2003. a. Ion occlusion/deocclusion partial reactions in individual palytoxin-modified Na/K pumps. Ann. N. Y. Acad. Sci. 986:116–126. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

