Death-receptor activation halts clathrin-dependent endocytosis
- PMID: 16801533
- PMCID: PMC1482799
- DOI: 10.1073/pnas.0604044103
Death-receptor activation halts clathrin-dependent endocytosis
Erratum in
- Proc Natl Acad Sci U S A. 2006 Sep 5;103(36):13560
-
Correction for Austin et al., Death-receptor activation halts clathrin-dependent endocytosis.Proc Natl Acad Sci U S A. 2025 Jan 28;122(4):e2425433121. doi: 10.1073/pnas.2425433121. Epub 2025 Jan 3. Proc Natl Acad Sci U S A. 2025. PMID: 39752512 Free PMC article. No abstract available.
Retraction in
-
Retraction for Austin et al., Death-receptor activation halts clathrin-dependent endocytosis.Proc Natl Acad Sci U S A. 2025 May 20;122(20):e2510913122. doi: 10.1073/pnas.2510913122. Epub 2025 May 15. Proc Natl Acad Sci U S A. 2025. PMID: 40372440 Free PMC article. No abstract available.
Abstract
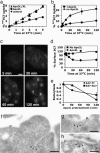
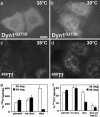
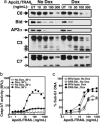
Endocytosis is crucial for various aspects of cell homeostasis. Here, we show that proapoptotic death receptors (DRs) trigger selective destruction of the clathrin-dependent endocytosis machinery. DR stimulation induced rapid, caspase-mediated cleavage of key clathrin-pathway components, halting cellular uptake of the classic cargo protein transferrin. DR-proximal initiator caspases cleaved the clathrin adaptor subunit AP2alpha between functionally distinct domains, whereas effector caspases processed clathrin's heavy chain. DR5 underwent ligand-induced, clathrin-mediated endocytosis, suggesting that internalization of DR signaling complexes facilitates clathrin-pathway targeting by caspases. An endocytosis-blocking, temperature-sensitive dynamin-1 mutant attenuated DR internalization, enhanced caspase stimulation downstream of DRs, and increased apoptosis. Thus, DR-triggered caspase activity disrupts clathrin-dependent endocytosis, leading to amplification of programmed cell death.
Conflict of interest statement
Conflict of interest statement: No conflicts declared.
Figures



Similar articles
-
TRAIL-death receptor endocytosis and apoptosis are selectively regulated by dynamin-1 activation.Proc Natl Acad Sci U S A. 2017 Jan 17;114(3):504-509. doi: 10.1073/pnas.1615072114. Epub 2017 Jan 3. Proc Natl Acad Sci U S A. 2017. PMID: 28049841 Free PMC article.
-
Protein kinase C modulates tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis by targeting the apical events of death receptor signaling.J Biol Chem. 2003 Nov 7;278(45):44338-47. doi: 10.1074/jbc.M307376200. Epub 2003 Aug 14. J Biol Chem. 2003. PMID: 12920112
-
Differential inhibition of TRAIL-mediated DR5-DISC formation by decoy receptors 1 and 2.Mol Cell Biol. 2006 Oct;26(19):7046-55. doi: 10.1128/MCB.00520-06. Mol Cell Biol. 2006. PMID: 16980609 Free PMC article.
-
Regulation of TRAIL-induced apoptosis by ectopic expression of antiapoptotic factors.Vitam Horm. 2004;67:453-83. doi: 10.1016/S0083-6729(04)67023-3. Vitam Horm. 2004. PMID: 15110190 Review.
-
Messengers of cell death: apoptotic signaling in health and disease.Haematologica. 2003 Feb;88(2):212-8. Haematologica. 2003. PMID: 12604411 Review.
Cited by
-
Monocyte chemotactic protein-induced protein-1 enhances DR5 degradation and negatively regulates DR5 activation-induced apoptosis through its deubiquitinase function.Oncogene. 2018 Jun;37(25):3415-3425. doi: 10.1038/s41388-018-0200-9. Epub 2018 Mar 19. Oncogene. 2018. PMID: 29551769
-
Hetero-modification of TRAIL trimer for improved drug delivery and in vivo antitumor activities.Sci Rep. 2015 Oct 8;5:14872. doi: 10.1038/srep14872. Sci Rep. 2015. PMID: 26445897 Free PMC article.
-
TRAIL-death receptor endocytosis and apoptosis are selectively regulated by dynamin-1 activation.Proc Natl Acad Sci U S A. 2017 Jan 17;114(3):504-509. doi: 10.1073/pnas.1615072114. Epub 2017 Jan 3. Proc Natl Acad Sci U S A. 2017. PMID: 28049841 Free PMC article.
-
The metabolite α-KG induces GSDMC-dependent pyroptosis through death receptor 6-activated caspase-8.Cell Res. 2021 Sep;31(9):980-997. doi: 10.1038/s41422-021-00506-9. Epub 2021 May 19. Cell Res. 2021. PMID: 34012073 Free PMC article.
-
The TRAIL to targeted therapy of breast cancer.Adv Cancer Res. 2009;103:43-73. doi: 10.1016/S0065-230X(09)03003-6. Adv Cancer Res. 2009. PMID: 19854352 Free PMC article. Review.
References
-
- Danial N. N., Korsmeyer S. J. Cell. 2004;116:205–219. - PubMed
-
- Strasser A., O’Connor L., Dixit V. M. Annu. Rev. Biochem. 2000;69:217–245. - PubMed
-
- Thornberry N. A., Lazebnik Y. Science. 1998;281:1312–1316. - PubMed
-
- Nagata S. Cell. 1997;88:355–365. - PubMed
-
- LeBlanc H. N., Ashkenazi A. Cell Death Differ. 2003;10:66–75. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

