Nongenomic oestrogen signalling in oestrogen receptor negative breast cancer cells: a role for the angiotensin II receptor AT1
- PMID: 16805920
- PMCID: PMC1557727
- DOI: 10.1186/bcr1509
Nongenomic oestrogen signalling in oestrogen receptor negative breast cancer cells: a role for the angiotensin II receptor AT1
Erratum in
-
Correction to: Nongenomic oestrogen signalling in oestrogen receptor negative breast cancer cells: a role for the angiotensin II receptor AT1.Breast Cancer Res. 2018 Jun 20;20(1):61. doi: 10.1186/s13058-018-0987-x. Breast Cancer Res. 2018. PMID: 29925428 Free PMC article.
Abstract
Introduction: Oestrogens can mediate some of their cell survival properties through a nongenomic mechanism that involves the mitogen-activated protein kinase (MAPK) pathway. The mechanism of this rapid signalling and its dependence on a membrane bound oestrogen receptor (ER), however, remains controversial. The role of G-protein-coupled receptor and epidermal growth factor (EGF) receptor in an ER-independent signalling pathway modulated by oestrogen was investigated.
Methods: ER-positive and ER-negative breast cancer cell lines (MCF-7 and SKBR3) and primary breast cancer cell cultures were used in this study. Cell proliferation was assessed using standard MTT assays. Protein and cAMP levels were detected by Western blotting and ELISA, respectively. Antigen localization was performed by immunocytochemistry, immunohistochemistry and immunofluorescence. Protein knockdown was achieved using small interfering RNA technologies.
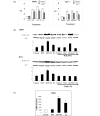
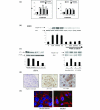
Results: EGF and oestrogen, alone and in combination, induced cell proliferation and phosphorylation of MAPK proteins Raf and ERK (extracellular signal regulated kinase)1/2 in both ER-negative SKBR3 and ER-positive MCF-7 human breast cancer cell lines. Increased Raf phosphorylation was also observed in primary human breast cultures derived from ER-positive and ER-negative breast tumours. Oestrogen induced an increase in intracellular cAMP in ER-negative SKBR3 human breast cancer cells. Oestrogen-mediated cell growth and phosphorylation of MAPK was modified by the EGF receptor antagonist AG1478, the G-protein antagonist pertussis toxin, and the angiotensin II receptor antagonist saralasin. Knockdown of angiotensin II type 1 receptor (AT1) protein expression with small interfering RNA attenuated oestrogen-induced Raf phosphorylation in ER-negative cells. AT1 receptor was found to be expressed in the cell membrane of breast tumour epithelial cells.
Conclusion: These findings provide evidence that, in breast cancer cells, oestrogen can signal through AT1 to activate early cell survival mechanisms in an ER-independent manner.
Figures


Similar articles
-
Estrogen action via the G protein-coupled receptor, GPR30: stimulation of adenylyl cyclase and cAMP-mediated attenuation of the epidermal growth factor receptor-to-MAPK signaling axis.Mol Endocrinol. 2002 Jan;16(1):70-84. doi: 10.1210/mend.16.1.0758. Mol Endocrinol. 2002. PMID: 11773440
-
Leptin induces upregulation of sphingosine kinase 1 in oestrogen receptor-negative breast cancer via Src family kinase-mediated, janus kinase 2-independent pathway.Breast Cancer Res. 2014 Oct 25;16(5):426. doi: 10.1186/s13058-014-0426-6. Breast Cancer Res. 2014. PMID: 25482303 Free PMC article.
-
Modulating therapeutic effects of the c-Src inhibitor via oestrogen receptor and human epidermal growth factor receptor 2 in breast cancer cell lines.Eur J Cancer. 2012 Dec;48(18):3488-98. doi: 10.1016/j.ejca.2012.04.020. Epub 2012 Jun 2. Eur J Cancer. 2012. PMID: 22658320 Free PMC article.
-
Inhibition of erbB receptor (HER) tyrosine kinases as a strategy to abrogate antiestrogen resistance in human breast cancer.Clin Cancer Res. 2001 Dec;7(12 Suppl):4436s-4442s; discussion 4411s-4412s. Clin Cancer Res. 2001. PMID: 11916237 Review.
-
Dynamic modelling of oestrogen signalling and cell fate in breast cancer cells.Nat Rev Cancer. 2011 Jun 16;11(7):523-32. doi: 10.1038/nrc3081. Nat Rev Cancer. 2011. PMID: 21677677 Free PMC article. Review.
Cited by
-
The renin-angiotensin system and cancer: old dog, new tricks.Nat Rev Cancer. 2010 Nov;10(11):745-59. doi: 10.1038/nrc2945. Epub 2010 Oct 22. Nat Rev Cancer. 2010. PMID: 20966920 Review.
-
Expression of estrogenicity genes in a lineage cell culture model of human breast cancer progression.Breast Cancer Res Treat. 2010 Feb;120(1):35-45. doi: 10.1007/s10549-009-0363-8. Epub 2009 Mar 24. Breast Cancer Res Treat. 2010. PMID: 19308726 Free PMC article.
-
Correction to: Nongenomic oestrogen signalling in oestrogen receptor negative breast cancer cells: a role for the angiotensin II receptor AT1.Breast Cancer Res. 2018 Jun 20;20(1):61. doi: 10.1186/s13058-018-0987-x. Breast Cancer Res. 2018. PMID: 29925428 Free PMC article.
-
ERK/CANP rapid signaling mediates 17β-estradiol-induced proliferation of human breast cancer cell line MCF-7 cells.Int J Clin Exp Med. 2014 Jan 15;7(1):156-62. eCollection 2014. Int J Clin Exp Med. 2014. PMID: 24482702 Free PMC article.
-
Estradiol stimulates transcription of nuclear respiratory factor-1 and increases mitochondrial biogenesis.Mol Endocrinol. 2008 Mar;22(3):609-22. doi: 10.1210/me.2007-0029. Epub 2007 Nov 29. Mol Endocrinol. 2008. PMID: 18048642 Free PMC article.
References
-
- Audy MC, Vacher P, Duly B. 17β-estradiol stimulates a rapid Ca2+ influx in LNCaP human prostate cancer cells. Eur J Endocrinol. 1996;135:367–373. - PubMed
-
- Nakhla AM, Khan MS, Rosner W. Biologically active steroids activate receptor-bound human sex hormone-binding globulin to cause LNCaP cells to accumulate adenosine 3', 5'-monophosphate. J Clin Endocrinol Metab. 1990;71:398–404. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

