A sensor for intracellular ionic strength
- PMID: 16815971
- PMCID: PMC1502282
- DOI: 10.1073/pnas.0603871103
A sensor for intracellular ionic strength
Abstract
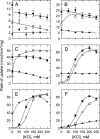
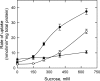
Cystathionine-beta-synthase (CBS) domains are found in >4,000 proteins in species from all kingdoms of life, yet their functions are largely unknown. Tandem CBS domains are associated with membrane transport proteins, most notably members of the ATP-binding cassette (ABC) superfamily; voltage-gated chloride channels and transporters; cation efflux systems; and various enzymes, transcription factors, and proteins of unknown function. We now show that tandem CBS domains in the osmoregulatory ABC transporter OpuA are sensors for ionic strength that control the transport activity through an electrostatic switching mechanism. The on/off state of the transporter is determined by the surface charge of the membrane and the internal ionic strength that is sensed by the CBS domains. By modifying the CBS domains, we can control the ionic strength dependence of the transporter: deleting a stretch of C-terminal anionic residues shifts the ionic strength dependence to higher values, whereas deleting the CBS domains makes the system largely independent of ionic strength. We present a model for the gating of membrane transport by ionic strength and propose a new role for CBS domains.
Conflict of interest statement
Conflict of interest statement: No conflicts declared.
Figures


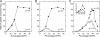

References
-
- Miles E. W., Kraus J. P. J. Biol. Chem. 2004;279:29871–29874. - PubMed
-
- Shan X., Kruger W. D. Nat. Genet. 1998;19:91–93. - PubMed
-
- Bateman A. Trends Biochem. Sci. 1997;22:12–13. - PubMed
-
- Zhang R., Evans G., Rotella F. J., Westbrook E. M., Beno D., Huberman E., Joachimiak A., Collart F. R. Biochemistry. 1999;38:4691–4700. - PubMed
-
- Miller M. D., Schwarzenbacher R., von Delft F., Abdubek P., Ambing E., Biorac T., Brinen L. S., Canaves J. M., Cambell J., Chiu H. J., et al. Proteins. 2004;57:213–217. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

