Naive CD4 T cells constitutively express CD40L and augment autoreactive B cell survival
- PMID: 16815973
- PMCID: PMC1484418
- DOI: 10.1073/pnas.0601539103
Naive CD4 T cells constitutively express CD40L and augment autoreactive B cell survival
Abstract
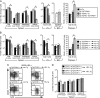
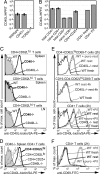
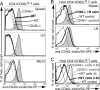
Chronic engagement of the B cell receptor by soluble autoantigen leads to reduced B cell survival. Using the Ig and hen egg lysozyme double transgenic mouse model, we demonstrate that the survival of soluble autoantigen-engaged B cells is further reduced in mice lacking CD4 T cells or deficient in CD40. Mixed bone marrow chimera experiments reveal that, under homeostatic conditions, the CD40L-CD40 pathway can augment autoreactive B cell survival in a non-cell-autonomous manner. Naive CD4 T cells are shown to constitutively express CD40L mRNA and protein, although cell surface CD40L abundance is low because of engagement with CD40 on other cells. These observations indicate that the CD40L-CD40 pathway can augment the survival of autoantigen-engaged B cells in the absence of T cell activation. We propose that constitutive CD40L expression by naive CD4 T cells influences the composition of the B cell repertoire and may also affect the homeostasis of other cell types such as regulatory T cells in lymphoid organs.
Conflict of interest statement
Conflict of interest statement: No conflicts declared.
Figures

References
-
- Goodnow C. C., Sprent J., Fazekas de St Groth B., Vinuesa C. G. Nature. 2005;435:590–597. - PubMed
-
- Wardemann H., Yurasov S., Schaefer A., Young J. W., Meffre E., Nussenzweig M. C. Science. 2003;301:1374–1377. - PubMed
-
- Cyster J. G., Goodnow C. C. Immunity. 1995;3:691–701. - PubMed
-
- Lesley R., Xu Y., Kalled S. L., Hess D. M., Schwab S. R., Shu H. B., Cyster J. G. Immunity. 2004;20:441–453. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

