Constitutive activation of two-component response regulators: characterization of VirG activation in Agrobacterium tumefaciens
- PMID: 16816192
- PMCID: PMC1539974
- DOI: 10.1128/JB.00387-06
Constitutive activation of two-component response regulators: characterization of VirG activation in Agrobacterium tumefaciens
Abstract
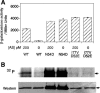
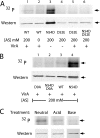
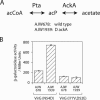
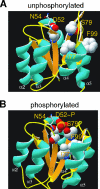
Response regulators are the ultimate modulators in two-component signal transduction pathways. The N-terminal receiver domains generally accept phosphates from cognate histidine kinases to control output. VirG for example, the response regulator of the VirA/VirG two-component system in Agrobacterium tumefaciens, mediates the expression of virulence genes in response to plant host signals. Response regulators have a highly conserved structure and share a similar conformational activation upon phosphorylation, yet the sequence and structural features that determine or perturb the cooperative activation events are ill defined. Here we use VirG and the unique features of the Agrobacterium system to extend our understanding of the response regulator activation. Two previously isolated constitutive VirG mutants, VirGN54D and VirGI77V/D52E, provide the foundation for our studies. In vivo phosphorylation patterns establish that VirGN54D is able to accumulate phosphates from small-molecule phosphate donors, such as acetyl phosphate, while the VirGI77V/D52E allele carries conformational changes mimicking the active conformation. Further structural alterations on these two alleles begin to reveal the changes necessary for response regulator activation.
Figures



Similar articles
-
Characterization of a virG mutation that confers constitutive virulence gene expression in Agrobacterium.Mol Microbiol. 1993 Feb;7(4):555-62. doi: 10.1111/j.1365-2958.1993.tb01146.x. Mol Microbiol. 1993. PMID: 8459774
-
Regulation of Agrobacterium tumefaciens virulence gene expression: isolation of a mutation that restores virGD52E function.Mol Plant Microbe Interact. 1995 Sep-Oct;8(5):788-91. doi: 10.1094/mpmi-8-0788. Mol Plant Microbe Interact. 1995. PMID: 7579624
-
Constitutive expression of the virulence genes improves the efficiency of plant transformation by Agrobacterium.Proc Natl Acad Sci U S A. 1994 Aug 2;91(16):7603-7. doi: 10.1073/pnas.91.16.7603. Proc Natl Acad Sci U S A. 1994. PMID: 8052627 Free PMC article.
-
Capturing the VirA/VirG TCS of Agrobacterium tumefaciens.Adv Exp Med Biol. 2008;631:161-77. doi: 10.1007/978-0-387-78885-2_11. Adv Exp Med Biol. 2008. PMID: 18792688 Review.
-
Host recognition by the VirA, VirG two-component regulatory proteins of agrobacterium tumefaciens.Res Microbiol. 1994 Jun-Aug;145(5-6):461-73. doi: 10.1016/0923-2508(94)90095-7. Res Microbiol. 1994. PMID: 7855433 Review.
Cited by
-
The atypical OmpR/PhoB response regulator ChxR from Chlamydia trachomatis forms homodimers in vivo and binds a direct repeat of nucleotide sequences.J Bacteriol. 2011 Jan;193(2):389-98. doi: 10.1128/JB.00833-10. Epub 2010 Nov 5. J Bacteriol. 2011. PMID: 21057008 Free PMC article.
-
Constitutive expression of the tzs gene from Agrobacterium tumefaciens virG mutant strains is responsible for improved transgenic plant regeneration in cotton meristem transformation.Plant Cell Rep. 2016 Mar;35(3):601-11. doi: 10.1007/s00299-015-1906-6. Epub 2015 Dec 9. Plant Cell Rep. 2016. PMID: 26650837
-
The aspartate-less receiver (ALR) domains: distribution, structure and function.PLoS Pathog. 2015 Apr 13;11(4):e1004795. doi: 10.1371/journal.ppat.1004795. eCollection 2015 Apr. PLoS Pathog. 2015. PMID: 25875291 Free PMC article.
-
5' and 3' Untranslated Regions Strongly Enhance Performance of Geminiviral Replicons in Nicotiana benthamiana Leaves.Front Plant Sci. 2016 Feb 24;7:200. doi: 10.3389/fpls.2016.00200. eCollection 2016. Front Plant Sci. 2016. PMID: 26941764 Free PMC article.
-
Use of restrained molecular dynamics to predict the conformations of phosphorylated receiver domains in two-component signaling systems.Proteins. 2017 Jan;85(1):155-176. doi: 10.1002/prot.25207. Epub 2016 Nov 20. Proteins. 2017. PMID: 27802580 Free PMC article.
References
-
- Birck, C., L. Mourey, P. Gouet, B. Fabry, J. Schumacher, P. Rousseau, D. Kahn, and J. P. Samama. 1999. Conformational changes induced by phosphorylation of the FixJ receiver domain. Structure 7:1505-1515. - PubMed
-
- Cangelosi, G. A., E. A. Best, G. Martinetti, and E. W. Nester. 1991. Genetic analysis of Agrobacterium. Methods Enzymol. 204:384-397. - PubMed
-
- Chamnongpol, S., and E. A. Groisman. 2000. Acetyl phosphate-dependent activation of a mutant PhoP response regulator that functions independently of its cognate sensor kinase. J. Mol. Biol. 300:291-305. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

