Role of HMGB1 in apoptosis-mediated sepsis lethality
- PMID: 16818669
- PMCID: PMC2118346
- DOI: 10.1084/jem.20052203
Role of HMGB1 in apoptosis-mediated sepsis lethality
Abstract
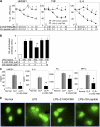
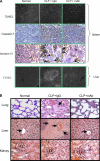
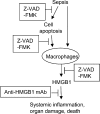
Severe sepsis, a lethal syndrome after infection or injury, is the third leading cause of mortality in the United States. The pathogenesis of severe sepsis is characterized by organ damage and accumulation of apoptotic lymphocytes in the spleen, thymus, and other organs. To examine the potential causal relationships of apoptosis to organ damage, we administered Z-VAD-FMK, a broad-spectrum caspase inhibitor, to mice with sepsis. We found that Z-VAD-FMK-treated septic mice had decreased levels of high mobility group box 1 (HMGB1), a critical cytokine mediator of organ damage in severe sepsis, and suppressed apoptosis in the spleen and thymus. In vitro, apoptotic cells activate macrophages to release HMGB1. Monoclonal antibodies against HMGB1 conferred protection against organ damage but did not prevent the accumulation of apoptotic cells in the spleen. Thus, our data indicate that HMGB1 production is downstream of apoptosis on the final common pathway to organ damage in severe sepsis.
Figures


References
-
- Angus, D., and R.S. Wax. 2001. Epidemiology of sepsis: an update. Crit. Care Med. 29:S109–S116. - PubMed
-
- Riedemann, N.C., R.-F. Guo, and P.A. Ward. 2003. Novel strategies for the treatment of sepsis. Nat. Med. 9:517–524. - PubMed
-
- Tracey, K.J. 2005. Fatal Sequence: the Killer Within. Dana Press, Washington, D.C. 224 pp.
-
- Lotze, M.T., and K.J. Tracey. 2005. High-mobility group box 1 protein (HMGB1): nuclear weapon in the immune arsenal. Nat. Rev. Immunol. 5:331–342. - PubMed
-
- Hotchkiss, R., K.C. Chang, P.E. Swanson, K.W. Tinsley, J.J. Hui, P. Klender, S. Xanthoudakis, S. Roy, C. Black, E. Grimm, et al. 2000. Caspase inhibitors improve survival in sepsis: a critical role of the lymphocyte. Nat. Immunol. 1:496–501. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

