IgD, like IgM, is a primordial immunoglobulin class perpetuated in most jawed vertebrates
- PMID: 16818885
- PMCID: PMC1636022
- DOI: 10.1073/pnas.0601407103
IgD, like IgM, is a primordial immunoglobulin class perpetuated in most jawed vertebrates
Abstract
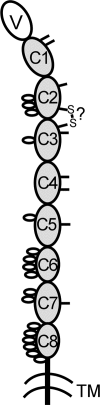
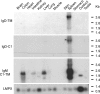
IgD has remained a mysterious Ig class and a bane to immunology students since its discovery >40 years ago. Its spotty occurrence in mammals and birds and the discovery of an isotype with similarities to IgD in bony fish are perplexing. We have identified IgD heavy (H) chain (delta) from the amphibian Xenopus tropicalis during examination of the IgH locus. The Xenopus delta gene is in the same position, immediately 3' of the IgM gene, as in mammals, and it is expressed only in the spleen at low levels, primarily as a transmembrane receptor by surface IgM(+) cells. Our data suggest that frog IgD is expressed on mature B cells, like in mouse/human. Unexpectedly, Xenopus IgD is orthologous to IgW, an Ig isotype found only in cartilaginous fish and lungfish, demonstrating that IgD/W, like IgM, was present in the ancestor of all living jawed vertebrates. In striking contrast to IgM, IgD/W is evolutionarily labile, showing many duplications/deletions of domains, the presence of multiple splice forms, existence as predominantly a secretory or transmembrane form, or loss of the entire gene in a species-specific manner. Our study suggests that IgD/W has played varied roles in different vertebrate taxa since the inception of the adaptive immune system, and it may have been preserved as a flexible locus over evolutionary time to complement steadfast IgM.
Conflict of interest statement
Conflict of interest statement: No conflicts declared.
Figures




References
-
- Flajnik M. F., Miller K., Du Pasquier L. In: Fundamental Immunology, 5th Ed. Paul W. E., editor. Philadelphia: Lippincott Williams & Wilkins; 2003. pp. 519–570.
-
- Kaattari S., Evans D., Klemer J. Immunol. Rev. 1998;166:133–142. - PubMed
-
- Preud’homme J. L., Petit I., Barra A., Morel F., Lecron J. C., Lelièvre E. Mol. Immunol. 2000;37:871–887. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources

