Microphthalmia-associated transcription factor interactions with 14-3-3 modulate differentiation of committed myeloid precursors
- PMID: 16822840
- PMCID: PMC1593166
- DOI: 10.1091/mbc.e06-05-0470
Microphthalmia-associated transcription factor interactions with 14-3-3 modulate differentiation of committed myeloid precursors
Abstract
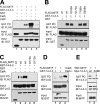
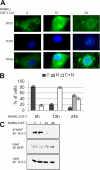
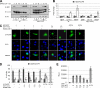
The microphthalmia-associated transcription factor (MITF) is required for terminal osteoclast differentiation and is a target for signaling pathways engaged by colony stimulating factor (CSF)-1 and receptor-activator of nuclear factor-kappaB ligand (RANKL). Work presented here demonstrates that MITF can shuttle from cytoplasm to nucleus dependent upon RANKL/CSF-1 action. 14-3-3 was identified as a binding partner of MITF in osteoclast precursors, and overexpression of 14-3-3 in a transgenic model resulted in increased cytosolic localization of MITF and decreased expression of MITF target genes. MITF/14-3-3 interaction was phosphorylation dependent, and Ser173 residue, within the minimal interaction region of amino acid residues 141-191, was required. The Cdc25C-associated kinase (C-TAK)1 interacted with an overlapping region of MITF. C-TAK1 increased MITF/14-3-3 complex formation and thus promoted cytoplasmic localization of MITF. C-TAK1 interaction was disrupted by RANKL/CSF-1 treatment. The results indicate that 14-3-3 regulates MITF activity by promoting the cytosolic localization of MITF in the absence of signals required for osteoclast differentiation. This work identifies a mechanism that regulates MITF activity in monocytic precursors that are capable of undergoing different terminal differentiation programs, and it provides a mechanism that allows committed precursors to rapidly respond to signals in the bone microenvironment to promote specifically osteoclast differentiation.
Figures


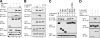
References
-
- Aitken A., Howell S., Jones D., Madrazo J., Martin H., Patel Y., Robinson K. Post-translationally modified 14-3-3 isoforms and inhibition of PKC. Mol. Cell Biochem. 1995;149–150:41–49. - PubMed
-
- Andrin C., Hendzel M. J. F-actin-dependent insolubility of chromatin-modifying components. J. Biol. Chem. 2004;279:25017–25023. - PubMed
-
- Bachmann M., Hennemann H., Xing P. X., Hoffmann I., Moroy T. The oncogenic serine/threonine kinase Pim-1 phosphorylates and inhibits the activity of Cdc25C-associated kinase 1 (C-TAK1): a novel role for Pim-1 at the G2/M cell cycle checkpoint. J. Biol. Chem. 2004;279:48319–48328. - PubMed
-
- Boyle W. J., Simonet W. S., Lacey D. L. Osteoclast differentiation and activation. Nature. 2003;423:337–342. - PubMed
-
- Brunet A., Bonni A., Zigmond M. J., Lin M. Z., Juo P., Hu L. S., Anderson M. J., Arden K. C., Blenis J., Greenberg M. E. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell. 1999;96:857–868. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

