Scotopic visual signaling in the mouse retina is modulated by high-affinity plasma membrane calcium extrusion
- PMID: 16822977
- PMCID: PMC1987386
- DOI: 10.1523/JNEUROSCI.5230-05.2006
Scotopic visual signaling in the mouse retina is modulated by high-affinity plasma membrane calcium extrusion
Abstract
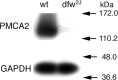
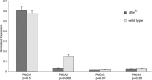
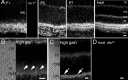
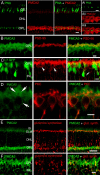
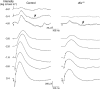
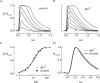
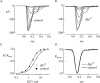
Transmission of visual signals at the first retinal synapse is associated with changes in calcium concentration in photoreceptors and bipolar cells. We investigated how loss of plasma membrane Ca2+ ATPase isoform 2 (PMCA2), the calcium transporter isoform with the highest affinity for Ca2+/calmodulin, affects transmission of rod- and cone-mediated responses. PMCA2 expression in the neuroblast layer was observed soon after birth; in the adult, PMCA2 was expressed in inner segments and synaptic terminals of rod photoreceptors, in rod bipolar cells, and in most inner retinal neurons but was absent from cones. To determine the role of PMCA2 in retinal signaling, we compared morphology and light responses of retinas from control mice and deafwaddler dfw2J mice, which lack functional PMCA2 protein. The cytoarchitecture of retinas from control and dfw2J mice was indistinguishable at the light microscope level. Suction electrode recordings revealed no difference in the sensitivity or amplitude of outer segment light responses of control and dfw2J rods. However, rod-mediated ERG b-wave responses in dfw2J mice were approximately 45% smaller and significantly slower than those of control mice. Furthermore, recordings from individual rod bipolar cells showed that the sensitivity of transmission at the rod output synapse was reduced by approximately 50%. No changes in the amplitude or timing of cone-mediated ERG responses were observed. These results suggest that PMCA2-mediated Ca2+ extrusion modulates the amplitude and timing of the high-sensitivity rod pathway to a much greater extent than that of the cone pathway.
Figures



Similar articles
-
Synaptic circuitry mediating light-evoked signals in dark-adapted mouse retina.Vision Res. 2004 Dec;44(28):3277-88. doi: 10.1016/j.visres.2004.07.045. Vision Res. 2004. PMID: 15535995
-
Genetic dissection of rod and cone pathways in the dark-adapted mouse retina.J Neurophysiol. 2009 Sep;102(3):1945-55. doi: 10.1152/jn.00142.2009. Epub 2009 Jul 8. J Neurophysiol. 2009. PMID: 19587322 Free PMC article.
-
Spatiotemporal regulation of ATP and Ca2+ dynamics in vertebrate rod and cone ribbon synapses.Mol Vis. 2007 Jun 15;13:887-919. Mol Vis. 2007. PMID: 17653034 Free PMC article.
-
Why rods and cones?Eye (Lond). 2016 Feb;30(2):179-85. doi: 10.1038/eye.2015.236. Epub 2015 Nov 13. Eye (Lond). 2016. PMID: 26563661 Free PMC article. Review.
-
Tuning outer segment Ca2+ homeostasis to phototransduction in rods and cones.Adv Exp Med Biol. 2002;514:179-203. doi: 10.1007/978-1-4615-0121-3_11. Adv Exp Med Biol. 2002. PMID: 12596922 Review.
Cited by
-
Development and maintenance of vision's first synapse.Dev Biol. 2021 Aug;476:218-239. doi: 10.1016/j.ydbio.2021.04.001. Epub 2021 Apr 10. Dev Biol. 2021. PMID: 33848537 Free PMC article. Review.
-
The dynamic architecture of photoreceptor ribbon synapses: cytoskeletal, extracellular matrix, and intramembrane proteins.Vis Neurosci. 2011 Nov;28(6):453-71. doi: 10.1017/S0952523811000356. Vis Neurosci. 2011. PMID: 22192503 Free PMC article. Review.
-
Store-Operated Calcium Entry in Müller Glia Is Controlled by Synergistic Activation of TRPC and Orai Channels.J Neurosci. 2016 Mar 16;36(11):3184-98. doi: 10.1523/JNEUROSCI.4069-15.2016. J Neurosci. 2016. PMID: 26985029 Free PMC article.
-
Trafficking of presynaptic PMCA signaling complexes in mouse photoreceptors requires Cav1.4 α1 subunits.Adv Exp Med Biol. 2012;723:739-44. doi: 10.1007/978-1-4614-0631-0_94. Adv Exp Med Biol. 2012. PMID: 22183401 Free PMC article. No abstract available.
-
Clock gene expression in gravid uterus and extra-embryonic tissues during late gestation in the mouse.Reprod Fertil Dev. 2010;22(5):743-50. doi: 10.1071/RD09243. Reprod Fertil Dev. 2010. PMID: 20450826 Free PMC article.
References
-
- Bayer AU, Mittag T, Cook P, Brodie SE, Podos SM, Maag KP (1999). Comparisons of the amplitude size and the reproducibility of three different electrodes to record the corneal flash electroretinogram in rodents. Doc Ophthalmol 98:233–246. - PubMed
-
- Berntson A, Smith RG, Taylor WR (2004). Postsynaptic calcium feedback between rods and rod bipolar cells in the mouse retina. Vis Neurosci 21:913–924. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
