Anti-CD20 monoclonal antibody with enhanced affinity for CD16 activates NK cells at lower concentrations and more effectively than rituximab
- PMID: 16825493
- PMCID: PMC1895597
- DOI: 10.1182/blood-2006-04-020057
Anti-CD20 monoclonal antibody with enhanced affinity for CD16 activates NK cells at lower concentrations and more effectively than rituximab
Abstract
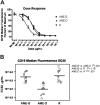
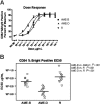
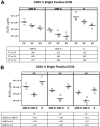
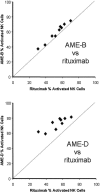
Growing evidence indicates that the affinity of monoclonal antibodies (mAbs) for CD16 (FcgammaRIII) plays a central role in the ability of the mAb to mediate antitumor activity. We evaluated how CD16 polymorphisms, and mAb with modified affinity for target antigen and CD16, affect natural killer (NK) cell phenotype when CD20(+) malignant B cells were also present. The mAb consisted of rituximab (R), anti-CD20 with enhanced affinity for CD20 (AME-B), and anti-CD20 with enhanced affinity for both CD20 and CD16 (AME-D). Higher concentrations of mAb were needed to induce CD16 modulation, CD54 up-regulation, and antibody-dependent cellular cytotoxicity (ADCC) on NK cells from subjects with the lower affinity CD16 polymorphism. The dose of mAb needed to induce NK activation was lower with AME-D irrespective of CD16 polymorphism. At saturating mAb concentrations, peak NK activation was greater for AME-D. Similar results were found with measurement of CD16 modulation, CD54 up-regulation, and ADCC. These data demonstrate that cells coated with mAb with enhanced affinity for CD16 are more effective at activating NK cells at both low and saturating mAb concentrations irrespective of CD16 polymorphism, and they provide further evidence for the clinical development of such mAbs with the goal of improving clinical response to mAb.
Figures



Similar articles
-
CD16 polymorphisms and NK activation induced by monoclonal antibody-coated target cells.J Immunol Methods. 2005 Sep;304(1-2):88-99. doi: 10.1016/j.jim.2005.06.018. J Immunol Methods. 2005. PMID: 16109421
-
Antibody Afucosylation Augments CD16-Mediated Serial Killing and IFNγ Secretion by Human Natural Killer Cells.Front Immunol. 2021 Mar 16;12:641521. doi: 10.3389/fimmu.2021.641521. eCollection 2021. Front Immunol. 2021. PMID: 33796107 Free PMC article.
-
Variable contribution of monoclonal antibodies to ADCC in patients with chronic lymphocytic leukemia.Leuk Lymphoma. 2009 Aug;50(8):1361-8. doi: 10.1080/10428190903026500. Leuk Lymphoma. 2009. PMID: 19562616
-
New anti-CD20 monoclonal antibodies for the treatment of B-cell lymphoid malignancies.BioDrugs. 2011 Feb 1;25(1):13-25. doi: 10.2165/11539590-000000000-00000. BioDrugs. 2011. PMID: 21090841 Review.
-
The future of CD20 monoclonal antibody therapy in B-cell malignancies.Leuk Lymphoma. 2010 Jun;51(6):983-94. doi: 10.3109/10428191003717746. Leuk Lymphoma. 2010. PMID: 20367564 Review.
Cited by
-
TriKEs and BiKEs join CARs on the cancer immunotherapy highway.Hum Vaccin Immunother. 2016 Nov;12(11):2790-2796. doi: 10.1080/21645515.2016.1198455. Epub 2016 Jun 20. Hum Vaccin Immunother. 2016. PMID: 27322989 Free PMC article.
-
Engineered NK Cells Against Cancer and Their Potential Applications Beyond.Front Immunol. 2022 Feb 15;13:825979. doi: 10.3389/fimmu.2022.825979. eCollection 2022. Front Immunol. 2022. PMID: 35242135 Free PMC article. Review.
-
γδ T-cell killing of primary follicular lymphoma cells is dramatically potentiated by GA101, a type II glycoengineered anti-CD20 monoclonal antibody.Haematologica. 2011 Mar;96(3):400-7. doi: 10.3324/haematol.2010.029520. Epub 2010 Nov 25. Haematologica. 2011. PMID: 21109686 Free PMC article.
-
Molecular impact of selective NFKB1 and NFKB2 signaling on DLBCL phenotype.Oncogene. 2017 Jul 20;36(29):4224-4232. doi: 10.1038/onc.2017.90. Epub 2017 Apr 3. Oncogene. 2017. PMID: 28368397
-
Rituximab: mechanism of action.Semin Hematol. 2010 Apr;47(2):115-23. doi: 10.1053/j.seminhematol.2010.01.011. Semin Hematol. 2010. PMID: 20350658 Free PMC article. Review.
References
-
- Byrd JC, Kitada S, Flinn IW, et al. The mechanism of tumor cell clearance by rituximab in vivo in patients with B-cell chronic lymphocytic leukemia: evidence of caspase activation and apoptosis induction. Blood. 2002;99: 1038-1043. - PubMed
-
- Jazirehi AR, Gan XH, De Vos S, Emmanouilides C, Bonavida B. Rituximab (anti-CD20) selectively modifies Bcl-xL and apoptosis protease activating factor-1 (Apaf-1) expression and sensitizes human non-Hodgkin's lymphoma B cell lines to paclitaxel-induced apoptosis. Mol Cancer Ther. 2003; 2: 1183-1193. - PubMed
-
- Golay J, Zaffaroni L, Vaccari T, et al. Biologic response of B lymphoma cells to anti-CD20 monoclonal antibody rituximab in vitro: CD55 and CD59 regulate complement-mediated cell lysis. Blood. 2000;95: 3900-3908. - PubMed
-
- Di Gaetano N, Cittera E, Nota R, et al. Complement activation determines the therapeutic activity of rituximab in vivo. J Immunol. 2003;171: 1581-1587. - PubMed
-
- Farag SS, Fehniger TA, Becknell B, Blaser BW, Caligiuri MA. New directions in natural killer cell-based immunotherapy of human cancer. Expert Opin Biol Ther. 2003;3: 237-250. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

