Genetic context determines susceptibility to intraocular pressure elevation in a mouse pigmentary glaucoma
- PMID: 16827931
- PMCID: PMC1543659
- DOI: 10.1186/1741-7007-4-20
Genetic context determines susceptibility to intraocular pressure elevation in a mouse pigmentary glaucoma
Abstract
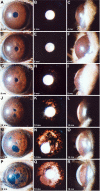
Background: DBA/2J (D2) mice develop an age-related form of glaucoma. Their eyes progressively develop iris pigment dispersion and iris atrophy followed by increased intraocular pressure (IOP) and glaucomatous optic nerve damage. Mutant alleles of the Gpnmb and Tyrp1 genes are necessary for the iris disease, but it is unknown whether alleles of other D2 gene(s) are necessary for the distinct later stages of disease. We initiated a study of congenic strains to further define the genetic requirements and disease mechanisms of the D2 glaucoma.
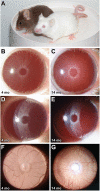
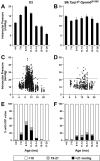
Results: To further understand D2 glaucoma, we created congenic strains of mice on the C57BL/6J (B6) genetic background. B6 double-congenic mice carrying D2-derived Gpnmb and Tyrp1 mutations develop a D2-like iris disease. B6 single-congenics with only the Gpnmb and Tyrp1 mutations develop milder forms of iris disease. Genetic epistasis experiments introducing a B6 tyrosinase mutation into the congenic strains demonstrated that both the single and double-congenic iris diseases are rescued by interruption of melanin synthesis. Importantly, our experiments analyzing mice at ages up to 27 months indicate that the B6 double-congenic mice are much less prone to IOP elevation and glaucoma than are D2 mice.
Conclusion: As demonstrated here, the Gpnmb and Tyrp1 iris phenotypes are both individually dependent on tyrosinase function. These results support involvement of abnormal melanosomal events in the diseases caused by each gene. In the context of the inbred D2 mouse strain, the glaucoma phenotype is clearly influenced by more genes than just Gpnmb and Tyrp1. Despite the outward similarity of pigment-dispersing iris disease between D2 and the B6 double-congenic mice, the congenic mice are much less susceptible to developing high IOP and glaucoma. These new congenic strains provide a valuable new resource for further studying the genetic and mechanistic complexity of this form of glaucoma.
Figures


References
-
- Allingham RR, Damji KF, Freedman S, Moroi SE, Shafranov G, Shields MB. Shields' Textbook of Glaucoma. 5. Philadelphia: Lippincott Williams and Wilkins;; 2004.
-
- Nickells RW, Jampel HD, Zack DJ. Glaucoma. In: Rimoin DL, Conner MJ, Pyeritz RE, Korf BR, editor. Emery & Rimoins Principles and Practices of Medical Genetics. 4. Vol. 3. Churchill Livingstone; 2002. pp. 3491–3512.
-
- Ritch R, Shields MB, Krupin T. The Glaucomas. St Louis Mosby; 1996.
-
- Palmberg PF, Wiggs JL. Mechanisms of glaucoma. In: Yanoff M, Duker JS, editor. Ophthalmology. 2. St Louis: Mosby; 2004.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

