Nf1+/- mast cells induce neurofibroma like phenotypes through secreted TGF-beta signaling
- PMID: 16835260
- PMCID: PMC3024714
- DOI: 10.1093/hmg/ddl165
Nf1+/- mast cells induce neurofibroma like phenotypes through secreted TGF-beta signaling
Abstract
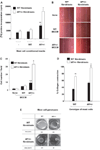
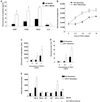
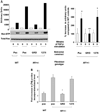
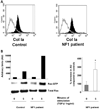
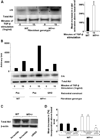
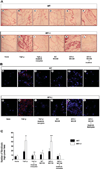
Neurofibromas are common tumors found in neurofibromatosis type 1 (NF1) patients. These complex tumors are composed of Schwann cells, mast cells, fibroblasts and perineurial cells embedded in collagen that provide a lattice for tumor invasion. Genetic studies demonstrate that in neurofibromas, nullizygous loss of Nf1 in Schwann cells and haploinsufficiency of Nf1 in non-neuronal cells are required for tumorigenesis. Fibroblasts are a major cellular constituent in neurofibromas and are a source of collagen that constitutes approximately 50% of the dry weight of the tumor. Here, we show that two of the prevalent heterozygous cells found in neurofibromas, mast cells and fibroblasts interact directly to contribute to tumor phenotype. Nf1+/- mast cells secrete elevated concentrations of the profibrotic transforming growth factor-beta (TGF-beta). In response to TGF-beta, both murine Nf1+/- fibroblasts and fibroblasts from human neurofibromas proliferate and synthesize excessive collagen, a hallmark of neurofibromas. We also establish that the TGF-beta response occurs via hyperactivation of a novel Ras-c-abl signaling pathway. Genetic or pharmacological inhibition of c-abl reverses fibroblast proliferation and collagen synthesis to wild-type levels. These studies identify a novel molecular target to inhibit neurofibroma formation.
Conflict of interest statement
Figures



Similar articles
-
Neurofibromin-deficient Schwann cells secrete a potent migratory stimulus for Nf1+/- mast cells.J Clin Invest. 2003 Dec;112(12):1851-61. doi: 10.1172/JCI19195. J Clin Invest. 2003. PMID: 14679180 Free PMC article.
-
Mast cells and the neurofibroma microenvironment.Blood. 2010 Jul 15;116(2):157-64. doi: 10.1182/blood-2009-09-242875. Epub 2010 Mar 16. Blood. 2010. PMID: 20233971 Free PMC article. Review.
-
Hyperactivation of p21(ras) and the hematopoietic-specific Rho GTPase, Rac2, cooperate to alter the proliferation of neurofibromin-deficient mast cells in vivo and in vitro.J Exp Med. 2001 Jul 2;194(1):57-69. doi: 10.1084/jem.194.1.57. J Exp Med. 2001. PMID: 11435472 Free PMC article.
-
Pak1 regulates multiple c-Kit mediated Ras-MAPK gain-in-function phenotypes in Nf1+/- mast cells.Blood. 2008 Dec 1;112(12):4646-54. doi: 10.1182/blood-2008-04-155085. Epub 2008 Sep 2. Blood. 2008. PMID: 18768391 Free PMC article.
-
Pathogenesis of plexiform neurofibroma: tumor-stromal/hematopoietic interactions in tumor progression.Annu Rev Pathol. 2012;7:469-95. doi: 10.1146/annurev-pathol-011811-132441. Epub 2011 Nov 7. Annu Rev Pathol. 2012. PMID: 22077553 Free PMC article. Review.
Cited by
-
The impact of host immune cells on the development of neurofibromatosis type 1: The abnormal immune system provides an immune microenvironment for tumorigenesis.Neurooncol Adv. 2019 Oct 18;1(1):vdz037. doi: 10.1093/noajnl/vdz037. eCollection 2019 May-Dec. Neurooncol Adv. 2019. PMID: 32642666 Free PMC article. Review.
-
Fibroblast diversity and plasticity in the tumor microenvironment: roles in immunity and relevant therapies.Cell Commun Signal. 2023 Sep 18;21(1):234. doi: 10.1186/s12964-023-01204-2. Cell Commun Signal. 2023. PMID: 37723510 Free PMC article. Review.
-
Nano-Drug Delivery Systems Targeting CAFs: A Promising Treatment for Pancreatic Cancer.Int J Nanomedicine. 2024 Mar 18;19:2823-2849. doi: 10.2147/IJN.S451151. eCollection 2024. Int J Nanomedicine. 2024. PMID: 38525013 Free PMC article. Review.
-
Insights into the Pathogenesis of NF1-Associated Neoplasms.JID Innov. 2021 Sep;1(3):100044. doi: 10.1016/j.xjidi.2021.100044. Epub 2021 Aug 20. JID Innov. 2021. PMID: 34604833 Free PMC article.
-
RAS Signaling Gone Awry in the Skin: The Complex Role of RAS in Cutaneous Neurofibroma Pathogenesis, Emerging Biological Insights.J Invest Dermatol. 2023 Aug;143(8):1358-1368. doi: 10.1016/j.jid.2023.01.043. Epub 2023 May 27. J Invest Dermatol. 2023. PMID: 37245145 Free PMC article. Review.
References
-
- Knudson AG. Two genetic his (more or less) to cancer. Nat. Rev. Cancer. 2001;2:157–162. - PubMed
-
- Cook WD, McCaw BJ. Accommodating haploinsufficient tumor suppressor genes in Knudson’s model. Oncogene. 2000;19:3434–3438. - PubMed
-
- Viskochil D, Buchberg AM, Xu G, Cawthon RM, Stevens J, Wolff RK, Culver M, Carey JC, Copeland NG, Jenkins NA, et al. Deletions and a translocation interrupt a cloned gene at the neurofibromatosis type 1 locus. Cell. 1990;62:187–192. - PubMed
-
- Wallace MR, Marchuk DA, Andersen LB, Letcher R, Odeh HM, Saulino AM, Fountain JW, Brereton A, Nicholson J, Mitchell AL, et al. Type 1 neurofibromatosis gene: identification of a large transcript disrupted in three NF1 patients. Science. 1990;249:181–186. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

