Structure function analysis of SH2D2A isoforms expressed in T cells reveals a crucial role for the proline rich region encoded by SH2D2A exon 7
- PMID: 16839418
- PMCID: PMC1553471
- DOI: 10.1186/1471-2172-7-15
Structure function analysis of SH2D2A isoforms expressed in T cells reveals a crucial role for the proline rich region encoded by SH2D2A exon 7
Abstract
Background: The activation induced T cell specific adapter protein (TSAd), encoded by SH2D2A, interacts with and modulates Lck activity. Several transcript variants of TSAd mRNA exist, but their biological significance remains unknown. Here we examined expression of SH2D2A transcripts in activated CD4+ T cells and used the SH2D2A variants as tools to identify functionally important regions of TSAd.
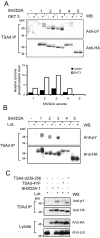
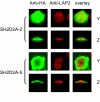
Results: TSAd was found to interact with Lck in human CD4+ T cells ex vivo. Three interaction modes of TSAd with Lck were identified. TSAd aa239-256 conferred binding to the Lck-SH3 domain, whereas one or more of the four tyrosines within aa239-334 encoded by SH2D2A exon 7 was found to confer interaction with the Lck-SH2-domain. Finally the TSAd-SH2 domain was found to interact with Lck. The SH2D2A exon 7 encoding TSAd aa 239-334 was found to harbour information essential not only for TSAd interaction with Lck, but also for TSAd modulation of Lck activity and translocation of TSAd to the nucleus. All five SH2D2A transcripts were found to be expressed in CD3 stimulated CD4+ T cells.
Conclusion: These data show that TSAd and Lck may interact through several different domains and that Lck TSAd interaction occurs in CD4+ T cells ex vivo. Alternative splicing of exon 7 encoding aa239-334 results in loss of the majority of protein interaction motives of TSAd and yields truncated TSAd molecules with altered ability to modulate Lck activity. Whether TSAd is regulated through differential alternative splicing of the SH2D2A transcript remains to be determined.
Figures

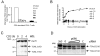


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

