Distinct ASIC currents are expressed in rat putative nociceptors and are modulated by nerve injury
- PMID: 16840516
- PMCID: PMC1995627
- DOI: 10.1113/jphysiol.2006.113035
Distinct ASIC currents are expressed in rat putative nociceptors and are modulated by nerve injury
Abstract
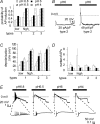
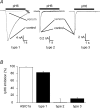
The H(+)-gated acid-sensing ion channels (ASICs) are expressed in dorsal root ganglion (DRG) neurones. Studies with ASIC knockout mice indicated either a pro-nociceptive or a modulatory role of ASICs in pain sensation. We have investigated in freshly isolated rat DRG neurones whether neurones with different ASIC current properties exist, which may explain distinct cellular roles, and we have investigated ASIC regulation in an experimental model of neuropathic pain. Small-diameter DRG neurones expressed three different ASIC current types which were all preferentially expressed in putative nociceptors. Type 1 currents were mediated by ASIC1a homomultimers and characterized by steep pH dependence of current activation in the pH range 6.8-6.0. Type 3 currents were activated in a similar pH range as type 1, while type 2 currents were activated at pH < 6. When activated by acidification to pH 6.8 or 6.5, the probability of inducing action potentials correlated with the ASIC current density. Nerve injury induced differential regulation of ASIC subunit expression and selective changes in ASIC function in DRG neurones, suggesting a complex reorganization of ASICs during the development of neuropathic pain. In summary, we describe a basis for distinct cellular functions of different ASIC types in small-diameter DRG neurones.
Figures




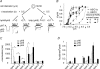


Similar articles
-
Proton sensitivity Ca2+ permeability and molecular basis of acid-sensing ion channels expressed in glabrous and hairy skin afferents.J Neurophysiol. 2006 Apr;95(4):2466-78. doi: 10.1152/jn.00861.2005. Epub 2006 Jan 11. J Neurophysiol. 2006. PMID: 16407431
-
Arachidonic acid potentiates acid-sensing ion channels in rat sensory neurons by a direct action.Neuroscience. 2007 Mar 16;145(2):686-98. doi: 10.1016/j.neuroscience.2006.12.024. Epub 2007 Jan 29. Neuroscience. 2007. PMID: 17258862
-
Acid-sensing ion channels ASIC2 and ASIC3 do not contribute to mechanically activated currents in mammalian sensory neurones.J Physiol. 2004 May 1;556(Pt 3):691-710. doi: 10.1113/jphysiol.2003.058693. Epub 2004 Feb 27. J Physiol. 2004. PMID: 14990679 Free PMC article.
-
Acid-sensing ion channels (ASICs): pharmacology and implication in pain.Pharmacol Ther. 2010 Dec;128(3):549-58. doi: 10.1016/j.pharmthera.2010.08.006. Epub 2010 Aug 31. Pharmacol Ther. 2010. PMID: 20807551 Review.
-
Peptides inhibitors of acid-sensing ion channels.Toxicon. 2007 Feb;49(2):271-84. doi: 10.1016/j.toxicon.2006.09.026. Epub 2006 Oct 4. Toxicon. 2007. PMID: 17113616 Review.
Cited by
-
Acid-sensing ion channels (ASICs) in mouse skeletal muscle afferents are heteromers composed of ASIC1a, ASIC2, and ASIC3 subunits.FASEB J. 2013 Feb;27(2):793-802. doi: 10.1096/fj.12-220400. Epub 2012 Oct 29. FASEB J. 2013. PMID: 23109675 Free PMC article.
-
Spider venom peptides as potential drug candidates due to their anticancer and antinociceptive activities.J Venom Anim Toxins Incl Trop Dis. 2019 Jun 3;25:e146318. doi: 10.1590/1678-9199-JVATITD-14-63-18. eCollection 2019. J Venom Anim Toxins Incl Trop Dis. 2019. PMID: 31210759 Free PMC article. Review.
-
Loss of acid-sensing ion channel 2 enhances pulmonary vascular resistance and hypoxic pulmonary hypertension.J Appl Physiol (1985). 2019 Aug 1;127(2):393-407. doi: 10.1152/japplphysiol.00894.2018. Epub 2019 Jun 6. J Appl Physiol (1985). 2019. PMID: 31169471 Free PMC article.
-
Deletion of annexin 2 light chain p11 in nociceptors causes deficits in somatosensory coding and pain behavior.J Neurosci. 2006 Oct 11;26(41):10499-507. doi: 10.1523/JNEUROSCI.1997-06.2006. J Neurosci. 2006. PMID: 17035534 Free PMC article.
-
T cell death-associated gene 8-mediated distinct signaling pathways modulate the early and late phases of neuropathic pain.iScience. 2024 Sep 13;27(10):110955. doi: 10.1016/j.isci.2024.110955. eCollection 2024 Oct 18. iScience. 2024. PMID: 39381739 Free PMC article.
References
-
- Akopian AN, Chen CC, Ding YN, Cesare P, Wood JN. A new member of the acid-sensing ion channel family. Neuroreport. 2000;11:2217–2222. - PubMed
-
- Baron A, Schaefer L, Lingueglia E, Champigny G, Lazdunski M. Zn2+ and H+ are coactivators of acid-sensing ion channels. J Biol Chem. 2001;276:35361–35367. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

