BK calcium-activated potassium channels regulate circadian behavioral rhythms and pacemaker output
- PMID: 16845385
- PMCID: PMC2909323
- DOI: 10.1038/nn1740
BK calcium-activated potassium channels regulate circadian behavioral rhythms and pacemaker output
Erratum in
- Nat Neurosci. 2006 Sep;9(9):1193
Abstract
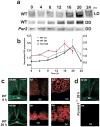
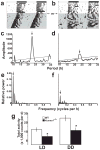
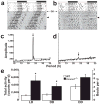
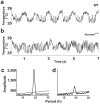
Spontaneous action potentials in the suprachiasmatic nucleus (SCN) are necessary for normal circadian timing of behavior in mammals. The SCN exhibits a daily oscillation in spontaneous firing rate (SFR), but the ionic conductances controlling SFR and the relationship of SFR to subsequent circadian behavioral rhythms are not understood. We show that daily expression of the large conductance Ca(2+)-activated K(+) channel (BK) in the SCN is controlled by the intrinsic circadian clock. BK channel-null mice (Kcnma1(-/-)) have increased SFRs in SCN neurons selectively at night and weak circadian amplitudes in multiple behaviors timed by the SCN. Kcnma1(-/-) mice show normal expression of clock genes such as Arntl (Bmal1), indicating a role for BK channels in SCN pacemaker output, rather than in intrinsic time-keeping. Our findings implicate BK channels as important regulators of the SFR and suggest that the SCN pacemaker governs the expression of circadian behavioral rhythms through SFR modulation.
Conflict of interest statement
The authors declare that they have no competing financial interests.
Figures

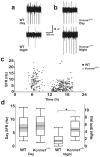
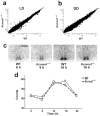
Comment in
-
BK channels and circadian output.Nat Neurosci. 2006 Aug;9(8):985-6. doi: 10.1038/nn0806-985. Nat Neurosci. 2006. PMID: 16871162 Free PMC article.
References
-
- King DP, Takahashi JS. Molecular genetics of circadian rhythms in mammals. Annu Rev Neurosci. 2000;23:713–742. - PubMed
-
- Reppert SM, Weaver DR. Coordination of circadian timing in mammals. Nature. 2002;418:935–941. - PubMed
-
- Hastings MH, Herzog ED. Clock genes, oscillators, and cellular networks in the suprachiasmatic nuclei. J Biol Rhythms. 2004;19:400–413. - PubMed
-
- Panda S, et al. Coordinated transcription of key pathways in the mouse by the circadian clock. Cell. 2002;109:307–320. - PubMed
-
- Ueda HR, et al. System-level identification of transcriptional circuits underlying mammalian circadian clocks. Nat Genet. 2005;37:187–192. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

