Cisplatin-induced toxicity is associated with platinum deposition in mouse kidney mitochondria in vivo and with selective inactivation of the alpha-ketoglutarate dehydrogenase complex in LLC-PK1 cells
- PMID: 16846239
- PMCID: PMC4133109
- DOI: 10.1021/bi060027g
Cisplatin-induced toxicity is associated with platinum deposition in mouse kidney mitochondria in vivo and with selective inactivation of the alpha-ketoglutarate dehydrogenase complex in LLC-PK1 cells
Abstract
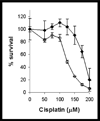
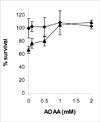
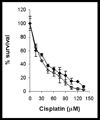
The anticancer drug cisplatin is nephrotoxic and neurotoxic. Previous data support the hypothesis that cisplatin is bioactivated to a nephrotoxicant. The final step in the proposed bioactivation is the formation of a platinum-cysteine S-conjugate followed by a pyridoxal 5'-phosphate (PLP)-dependent cysteine S-conjugate beta-lyase reaction. This reaction would generate pyruvate, ammonium, and a highly reactive platinum (Pt)-thiol compound in vivo that would bind to proteins. In this work, the cellular location and identity of the PLP-dependent cysteine S-conjugate beta-lyase were investigated. Pt was shown to bind to proteins in kidneys of cisplatin-treated mice. The concentration of Pt-bound proteins was higher in the mitochondrial fraction than in the cytosolic fraction. Treatment of the mice with aminooxyacetic acid (AOAA, a PLP enzyme inhibitor), which had previously been shown to block the nephrotoxicity of cisplatin, decreased the binding of Pt to mitochondrial proteins but had no effect on the amount of Pt bound to proteins in the cytosolic fraction. These data indicate that a mitochondrial enzyme catalyzes the PLP-dependent cysteine S-conjugate beta-lyase reaction. PLP-dependent mitochondrial aspartate aminotransferase (mitAspAT) is a mitochondrial enzyme that catalyzes beta-elimination reactions with cysteine S-conjugates of halogenated alkenes. We reasoned that the enzyme might also catalyze a beta-lyase reaction with the cisplatin-cysteine S-conjugate. In this study, mitAspAT was stably overexpressed in LLC-PK(1) cells. Cisplatin was significantly more toxic in confluent monolayers of LLC-PK(1) cells that overexpressed mitAspAT than in control cells containing vector alone. AOAA completely blocked the cisplatin toxicity in confluent mitAspAT-transfected cells. The Pt-thiol compound could rapidly bind proteins and inactivate enzymes in close proximity of the PLP-dependent cysteine S-conjugate beta-lyase. Treatment with 50 or 100 microM cisplatin for 3 h, followed by removal of cisplatin from the medium for 24 h, resulted in a pronounced loss of alpha-ketoglutarate dehydrogenase complex (KGDHC) activity in both mitAspAT-transfected cells and control cells. Exposure to 100 microM cisplatin resulted in a significantly greater loss of KGDHC activity in the cells overexpressing mitAspAT than in control cells. Aconitase activity was diminished in both cell types, but only at the higher level of exposure to cisplatin. AspAT activity was also significantly decreased by cisplatin treatment. By contrast, several other enzymes (both cytosolic and mitochondrial) involved in energy/amino acid metabolism were not significantly affected by cisplatin treatment in the LLC-PK(1) cells, whether or not mitAspAT was overexpressed. The susceptibility of KGDHC and aconitase to inactivation in kidney cells exposed to cisplatin metabolites may be due to the proximity of mitAspAT to KGDHC and aconitase in mitochondria. These findings support the hypothesis that a mitochondrial cysteine S-conjugate beta-lyase converts the cisplatin-cysteine S-conjugate to a toxicant, and the data are consistent with the hypothesis that mitAspAT plays a role in the bioactivation of cisplatin.
Figures



References
-
- Masters JR, Koberle B. Curing metastatic cancer: lessons from testicular germ-cell tumours. Nat. Rev. Cancer. 2003;3:517–525. - PubMed
-
- Arany I, Safirstein RL. Cisplatin nephrotoxicity. Semin. Nephrol. 2003;23:460–464. - PubMed
-
- Steeghs N, de Jongh FE, Sillevis Smitt PA, van den Bent MJ. Cisplatin-induced encephalopathy and seizures. Anticancer Drugs. 2003;14:443–446. - PubMed
-
- Siddik ZH. Cisplatin: mode of cytotoxic action and molecular basis of resistance. Oncogene. 2003;22:7265–7279. - PubMed
-
- Boulikas T, Vougiouka M. Cisplatin and platinum drugs at the molecular level (review) Oncology Reports. 2003;10:1663–1682. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

