A phase I dose-finding and pharmacokinetic study of subcutaneous semisynthetic homoharringtonine (ssHHT) in patients with advanced acute myeloid leukaemia
- PMID: 16847470
- PMCID: PMC2360653
- DOI: 10.1038/sj.bjc.6603265
A phase I dose-finding and pharmacokinetic study of subcutaneous semisynthetic homoharringtonine (ssHHT) in patients with advanced acute myeloid leukaemia
Abstract
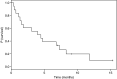
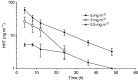
To determine the maximum-tolerated dose (MTD), dose-limiting toxicities and pharmacokinetic of semisynthetic homoharringtonine (ssHHT), given as a twice daily subcutaneous (s.c.) injections for 9 days, in patients with advanced acute leukaemia, 18 patients with advanced acute myeloid leukaemia were included in this sequential Bayesian phase I dose-finding trial. A starting dose of 0.5 mg m(-2) day(-1) was explored with subsequent dose escalations of 1, 3, 5 and 6 mg m(-2) day(-1). Myelosuppression was constant. The MTD was estimated as the dose level of 5 mg m(-2) day(-1) for 9 consecutive days by s.c. route. Dose-limiting toxicities were hyperglycaemia with hyperosmolar coma at 3 mg m(-2), and (i) one anasarque and haematemesis, (ii) one life-threatening pulmonary aspergillosis, (iii) one skin rash and (iv) one scalp pain at dose level of 5 mg m(-2) day(-1). The mean half-life of ssHHT was 11.01+/-3.4 h, the volume of distribution at steady state was 2+/-1.4 l kg(-1) and the plasma clearance was 11.6+/-10.4 l h(-1). Eleven of the 12 patients with circulating leukaemic cells had blood blast clearance, two achieved complete remission and one with blast crisis of CMML returned in chronic phase. The recommended daily dose of ssHHT on the 9-day schedule is 5 mg m(-2) day(-1).
Figures
Similar articles
-
Phase I/II study of subcutaneous homoharringtonine in patients with chronic myeloid leukemia who have failed prior therapy.Cancer. 2007 Jan 15;109(2):248-55. doi: 10.1002/cncr.22398. Cancer. 2007. PMID: 17154172 Clinical Trial.
-
A phase I clinical trial of the histone deacetylase inhibitor belinostat in patients with advanced hematological neoplasia.Eur J Haematol. 2008 Sep;81(3):170-6. doi: 10.1111/j.1600-0609.2008.01102.x. Epub 2008 May 27. Eur J Haematol. 2008. PMID: 18510700 Clinical Trial.
-
Phase I study of cladribine, cytarabine (Ara-C), granulocyte colony stimulating factor (G-CSF) (CLAG Regimen) and simultaneous escalating doses of imatinib mesylate (Gleevec) in relapsed/refractory AML.Leuk Res. 2008 Dec;32(12):1830-6. doi: 10.1016/j.leukres.2008.04.026. Epub 2008 Jun 20. Leuk Res. 2008. PMID: 18571721 Clinical Trial.
-
Phase I clinical investigation of homoharringtonine.Cancer Treat Rep. 1984 Sep;68(9):1085-91. Cancer Treat Rep. 1984. PMID: 6478448
-
Phase I trial of homoharringtonine in children with refractory leukemia.Cancer Treat Rep. 1987 Dec;71(12):1245-8. Cancer Treat Rep. 1987. PMID: 3480043
Cited by
-
Homoharringtonine reduced Mcl-1 expression and induced apoptosis in chronic lymphocytic leukemia.Blood. 2011 Jan 6;117(1):156-64. doi: 10.1182/blood-2010-01-262808. Epub 2010 Oct 22. Blood. 2011. PMID: 20971952 Free PMC article.
-
Drug monographs: cabozantanib and omacetaxine.Hosp Pharm. 2013 May;48(5):373-7. doi: 10.1310/hpj4805-373. Hosp Pharm. 2013. PMID: 24421492 Free PMC article.
-
Challenges in implementing model-based phase I designs in a grant-funded clinical trials unit.Trials. 2017 Dec 28;18(1):620. doi: 10.1186/s13063-017-2389-2. Trials. 2017. PMID: 29282111 Free PMC article.
-
Homoharringtonine: mechanisms, clinical applications and research progress.Front Oncol. 2025 Jan 30;15:1522273. doi: 10.3389/fonc.2025.1522273. eCollection 2025. Front Oncol. 2025. PMID: 39949739 Free PMC article. Review.
-
Omacetaxine mepesuccinate in the treatment of intractable chronic myeloid leukemia.Onco Targets Ther. 2014 Jan 31;7:177-86. doi: 10.2147/OTT.S41786. eCollection 2014. Onco Targets Ther. 2014. PMID: 24516334 Free PMC article. Review.
References
-
- Baguley BC, Calveley SB, Crowe KK, Fray LM, O'Rourke SA, Smith GP (1989) Comparison of the effects of flavone acetic acid, fostriecin, homoharringtonine and tumour necrosis factor alpha on colon 38 tumours in mice. Eur J Cancer Clin Oncol 25: 263–269 - PubMed
-
- Bennett JM, Young ML, Andersen JW, Cassileth PA, Tallman MS, Paietta E, Wiernik PH, Rowe JM (1997) Long-term survival in acute myeloid leukemia: the Eastern Cooperative Oncology Group experience. Cancer 80: 2205–2209 - PubMed
-
- Buchner T, Hiddemann W, Schoch C, Haferlach T, Sauerland MC, Heinecke A (2001) Acute myeloid leukaemia (AML): treatment of the older patient. Best Pract Res Clin Haematol 14: 139–151 - PubMed
-
- Cai Z, Lin M, Wuchter C, Ruppert V, Dorken B, Ludwig WD, Karawajew L (2001) Apoptotic response to homoharringtonine in human wt p53 leukemic cells is independent of reactive oxygen species generation and implicates Bax translocation, mitochondrial cytochrome c release and caspase activation. Leukemia 15: 567–574 - PubMed
-
- Cheson BD, Cassileth PA, Head DR, Schiffer CA, Bennett JM, Bloomfield CD, Brunning R, Gale RP, Grever MR, Keating MJ, Sawitsky A, Stass S, Weinstein H, Woods WG (1990) Report of the National Cancer Institute-sponsored workshop on definitions of diagnosis and response in acute myeloid leukemia. J Clin Oncol 8: 813–819 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

