Lewis x antigen mediates adhesion of human breast carcinoma cells to activated endothelium. Possible involvement of the endothelial scavenger receptor C-type lectin
- PMID: 16850248
- PMCID: PMC2288708
- DOI: 10.1007/s10549-006-9286-9
Lewis x antigen mediates adhesion of human breast carcinoma cells to activated endothelium. Possible involvement of the endothelial scavenger receptor C-type lectin
Abstract
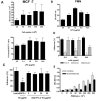
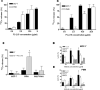
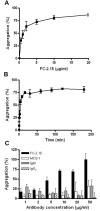
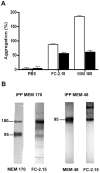
Lewis x (Le(x), CD15), also known as SSEA-1 (stage specific embryonic antigen-1), is a trisaccharide with the structure Galbeta(1-4)Fucalpha(1-3)GlcNAc, which is expressed on glycoconjugates in human polymorphonuclear granulocytes and various tumors such as colon and breast carcinoma. We have investigated the role of Le(x) in the adhesion of MCF-7 human breast cancer cells and PMN to human umbilical endothelial cells (HUVEC) and the effects of two different anti-Le(x) mAbs (FC-2.15 and MCS-1) on this adhesion. We also analyzed the cytolysis of Le(x+)-cells induced by anti-Le(x) mAbs and complement when cells were adhered to the endothelium, and the effect of these antibodies on HUVEC. The results indicate that MCF-7 cells can bind to HUVEC, and that MCS-1 but not FC-2.15 mAb inhibit this interaction. Both mAbs can efficiently lyse MCF-7 cells bound to HUVEC in the presence of complement without damaging endothelial cells. We also found a Le(x)-dependent PMN interaction with HUVEC. Although both anti-Le(x) mAbs lysed PMN in suspension and adhered to HUVEC, PMN aggregation was only induced by mAb FC-2.15. Blotting studies revealed that the endothelial scavenger receptor C-type lectin (SRCL), which binds Le(x)-trisaccharide, interacts with specific glycoproteins of M (r ) approximately 28 kD and 10 kD from MCF-7 cells. The interaction between Le(x+)-cancer cells and vascular endothelium is a potential target for cancer treatment.
Figures


Similar articles
-
Differential lytic and agglutinating activity of the anti-Lewis(x) monoclonal antibody FC-2.15 on human polymorphonuclear neutrophils and MCF-7 breast tumor cells. In vitro and ex vivo studies.Cancer Immunol Immunother. 1999 May-Jun;48(2-3):100-8. doi: 10.1007/s002620050553. Cancer Immunol Immunother. 1999. PMID: 10414463 Free PMC article.
-
Reactivity of monoclonal antibody FC-2.15 against drug resistant breast cancer cells. Additive cytotoxicity of adriamycin and taxol with FC-2.15.Breast Cancer Res Treat. 1998 Jan;47(2):163-70. doi: 10.1023/a:1005949118919. Breast Cancer Res Treat. 1998. PMID: 9497104
-
Monoclonal antibodies to the carbohydrate structure Lewis(x) stimulate the adhesive activity of leukocyte integrin CD11b/CD18 (CR3, Mac-1, alpha m beta 2) on human granulocytes.J Leukoc Biol. 1993 May;53(5):541-9. doi: 10.1002/jlb.53.5.541. J Leukoc Biol. 1993. PMID: 8099115
-
Dimethyl sulfoxide (DMSO) increases expression of sialyl Lewis x antigen and enhances adhesion of human gastric carcinoma (NUGC4) cells to activated endothelial cells.Int J Cancer. 1993 May 8;54(2):296-301. doi: 10.1002/ijc.2910540222. Int J Cancer. 1993. PMID: 7683631
-
Neutrophil adhesion to TNF alpha-activated endothelial cells potentiates leukotriene B4 production.J Cell Physiol. 1992 Oct;153(1):187-95. doi: 10.1002/jcp.1041530123. J Cell Physiol. 1992. PMID: 1381715
Cited by
-
The Role of Tumor-Associated Neutrophils in Early Luminal HER2-Negative Breast Cancer Progression.Asian Pac J Cancer Prev. 2025 Jan 1;26(1):207-213. doi: 10.31557/APJCP.2025.26.1.207. Asian Pac J Cancer Prev. 2025. PMID: 39874003 Free PMC article.
-
Rapid and Efficient Purification of Functional Collectin-12 and Its Opsonic Activity against Fungal Pathogens.J Immunol Res. 2019 Jul 29;2019:9164202. doi: 10.1155/2019/9164202. eCollection 2019. J Immunol Res. 2019. PMID: 31482100 Free PMC article.
-
Prognostic and Therapeutic Role of CD15 and CD15s in Cancer.Cancers (Basel). 2022 Apr 28;14(9):2203. doi: 10.3390/cancers14092203. Cancers (Basel). 2022. PMID: 35565333 Free PMC article. Review.
-
Invasive breast cancer in Argentine women: association between risk and prognostic factors with antigens of a peptidic and carbohydrate nature.Breast Cancer (Dove Med Press). 2011 Dec 20;3:161-73. doi: 10.2147/BCTT.S26833. eCollection 2011. Breast Cancer (Dove Med Press). 2011. PMID: 24367185 Free PMC article.
-
Fucosylated Antigens in Cancer: An Alliance toward Tumor Progression, Metastasis, and Resistance to Chemotherapy.Front Oncol. 2018 Feb 23;8:39. doi: 10.3389/fonc.2018.00039. eCollection 2018. Front Oncol. 2018. PMID: 29527514 Free PMC article. Review.
References
-
- Gooi HC, Feizi T, Kapadia A, Knowles BB, Solter D, Evans MJ. Stage-specific embryonic antigen involves α1→3 fucosylated type 2 blood group chains. Nature. 1981;292:156–158. - PubMed
-
- Hakomori S, Nudelman E, Levery S, Solter D, Knowles BB. The hapten structure of a developmentally regulated glycolipid antigen (SSEA-1) isolated from human erytrocytes and adenocarcinoma: a preliminary note. Biochem Biophys Res Commun. 1981;100:1578–1586. - PubMed
-
- Spooncer E, Fukuda M, Klock JC, Oates JE, Dell A. Isolation and characterization of polyfucosylated lactosaminoglycan from human granulocytes. J Biol Chem. 1984;259:4792–4801. - PubMed
-
- Mordoh J, Leis S, Bravo AI, Podhajcer OL, Ballaré C, Capurro M, Kairiyama C, Bover L. Description of a new monoclonal antibody, FC-2.15, reactive with human breast cancer and other human neoplasia. Int J Biological Markers. 1994;9:125–134. - PubMed
-
- Hittelet A, Camby I, Nagy N, Legendre H, Bronckart Y, Decaestecker C, Kaltner H, Nifant'ev NE, Bovin NV, Pector JC, Salmon I, Gabius HJ, Kiss R, Yeaton P. Binding sites for Lewis antigens are expressed by human colon cancer cells and negatively affect their migration. Lab Invest. 2003;83:777–787. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

