Infection with Porcine reproductive and respiratory syndrome virus stimulates an early gamma interferon response in the serum of pigs
- PMID: 16850939
- PMCID: PMC1477926
Infection with Porcine reproductive and respiratory syndrome virus stimulates an early gamma interferon response in the serum of pigs
Abstract
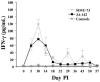
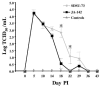
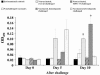
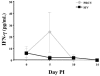
The early release of cytokines by cells involved in innate immunity is an important host response to intracellular pathogens. Gamma interferon (IFN-gamma) is an important cytokine produced during the early stages of an infection by macrophages, natural killer (NK) cells, and other cell types, and it is also a central cytokine mediator for the induction of cellular or Th1 immunity. To better understand innate and adaptive immune responses after infection with Porcine reproductive and respiratory syndrome virus (PRRSV), we investigated serum IFN-gamma concentrations and the duration of viremia. For 2 strains of atypical PRRSV, IFN-gamma was detectable in swine serum soon after infection and lasted for approximately 3 wk. Serum concentrations of IFN-gamma peaked at about 10 d after inoculation and returned to approximately baseline levels by day 22. However, individual pigs manifested short, sporadic increases in the serum concentration of IFN-gamma from 18 to 50 d after inoculation. Prior vaccination blocked the serum IFN-gamma response associated with homologous virus challenge and altered the kinetics of the response after heterologous challenge. Two other respiratory viruses of pigs, Porcine respiratory coronavirus and Swine influenza virus, do not appear to induce serum IFN-gamma. The early production of IFN-gamma in PRRSV-infected pigs might result from activation of NK cells, a response that is more characteristic of immune pathways stimulated by intracellular bacterial and protozoan infections.
La réponse hâtive en cytokines par les cellules impliquées dans l’immunité innée joue un rôle important dans la réponse envers les agents pathogènes intracellulaires. L’interféron gamma (IFN-γ) est une cytokine importante produite dans les premiers stages d’une infection par les macrophages, par les cellules tueuses naturelles (NK), ainsi que par d’autres types cellulaires, et elle est également une cytokine centrale médiatrice pour l’induction d’une réponse cellulaire ou immunité Th1. Afin de mieux comprendre les réponses immunitaires innées et acquises suite à l’infection par le virus du syndrome reproducteur et respiratoire porcin (PRRSV), nous avons étudié les concentrations sériques d’IFN-γ et la durée de la virémie. Pour deux souches atypiques de PRRSV, la présence d’IFN-γ était détectable dans le sérum de porc tôt après l’infection et dura environ 3 sem. Les concentrations sériques d’IFN-γ atteignirent leur maximum environ 10 j après l’inoculation et revinrent aux alentours du niveau de base au jour 22. Toutefois, quelques animaux présentèrent des augmentations de concentration sérique d’IFN-γ sporadiques de courte durée du jour 18 au jour 50 après l’inoculation. Une vaccination préalable empêcha la réponse sérique en IFN-γ associée à un test de provocation avec la souche homologue et modifia la cinétique de la réponse à un test de provocation avec une souche hétérologue. Deux autres virus respiratoires porcins, le virus de l’influenza porcin et le coronavirus respiratoire porcin, ne semblent pas induire la production d’IFN-γ. La production hâtive d’IFN-γ chez des porcs infectés par le PRRSV pourrait résulter d’une activation des cellules NK, une réponse qui est plutôt caractéristique des mécanismes immunitaires stimulés par les infections par les bactéries intracellulaires et les protozoaires.
(Traduit par Docteur Serge Messier)
Figures
Similar articles
-
The effect of a killed porcine reproductive and respiratory syndrome virus (PRRSV) vaccine treatment on virus shedding in previously PRRSV infected pigs.Vet Microbiol. 2004 Aug 19;102(1-2):11-8. doi: 10.1016/j.vetmic.2004.05.006. Vet Microbiol. 2004. PMID: 15288922
-
Efficacy of a modified live porcine reproductive and respiratory syndrome virus (PRRSV) vaccine in pigs naturally exposed to a heterologous European (Italian cluster) field strain: Clinical protection and cell-mediated immunity.Vaccine. 2009 Jun 8;27(28):3788-99. doi: 10.1016/j.vaccine.2009.03.028. Epub 2009 Apr 3. Vaccine. 2009. PMID: 19442420
-
Lymphocyte activation as cytokine gene expression and secretion is related to the porcine reproductive and respiratory syndrome virus (PRRSV) isolate after in vitro homologous and heterologous recall of peripheral blood mononuclear cells (PBMC) from pigs vaccinated and exposed to natural infection.Vet Immunol Immunopathol. 2013 Feb 15;151(3-4):193-206. doi: 10.1016/j.vetimm.2012.11.006. Epub 2012 Nov 19. Vet Immunol Immunopathol. 2013. PMID: 23228653
-
Interferon-gamma: biologic functions and HCV therapy (type I/II) (1 of 2 parts).Clin Ter. 2006 Jul-Aug;157(4):377-86. Clin Ter. 2006. Retraction in: Clin Ter. 2008 May-Jun;159(3):207. PMID: 17051976 Retracted. Review.
-
Proinflammatory cytokines and viral respiratory disease in pigs.Vet Res. 2000 Mar-Apr;31(2):187-213. doi: 10.1051/vetres:2000113. Vet Res. 2000. PMID: 10779199 Review.
Cited by
-
Cytokine profiles in pregnant gilts experimentally infected with porcine reproductive and respiratory syndrome virus and relationships with viral load and fetal outcome.Vet Res. 2014 Dec 6;45:113. doi: 10.1186/s13567-014-0113-8. Vet Res. 2014. PMID: 25479904 Free PMC article.
-
Oral Bacillus subtilis spores-based vaccine for mass vaccination against porcine reproductive and respiratory syndrome.Sci Rep. 2024 Nov 12;14(1):27742. doi: 10.1038/s41598-024-79387-y. Sci Rep. 2024. PMID: 39533088 Free PMC article.
-
Mannan oligosaccharide increases serum concentrations of antibodies and inflammatory mediators in weanling pigs experimentally infected with porcine reproductive and respiratory syndrome virus.J Anim Sci. 2012 Aug;90(8):2784-93. doi: 10.2527/jas.2011-4518. Epub 2012 Feb 24. J Anim Sci. 2012. PMID: 22367071 Free PMC article.
-
The influence of experimental infection of gilts with swine H1N2 influenza A virus during the second month of gestation on the course of pregnancy, reproduction parameters and clinical status.BMC Vet Res. 2014 Jun 4;10:123. doi: 10.1186/1746-6148-10-123. BMC Vet Res. 2014. PMID: 24893655 Free PMC article. Clinical Trial.
-
The 15N and 46R Residues of Highly Pathogenic Porcine Reproductive and Respiratory Syndrome Virus Nucleocapsid Protein Enhance Regulatory T Lymphocytes Proliferation.PLoS One. 2015 Sep 23;10(9):e0138772. doi: 10.1371/journal.pone.0138772. eCollection 2015. PLoS One. 2015. PMID: 26397116 Free PMC article.
References
-
- Cavanagh D. Nidovirales: a new order comprising Coronaviridae and Arteriviridae. Arch Virol. 1997;142:629–633. - PubMed
-
- Benfield DA, Collins JE, Dee SA, et al. Porcine reproductive and respiratory syndrome. In: Straw BE, D’Allaire S, Mengeling WL, Taylor DJ, eds. Diseases of Swine. Ames, Iowa: Iowa State University Press, 1999:201–232.
-
- Labarque GG, Nauwynck HJ, Van Reeth K, Penseart MB. Effect of cellular changes and onset of humoral immunity on the replication of porcine reproductive and respiratory syndrome virus in the lungs of pigs. J Gen Virol. 2000;81:1327–1334. - PubMed
-
- Mengeling WL, Lager KM, Vorwald AC. Diagnosis of porcine reproductive and respiratory syndrome. J Vet Diagn Invest. 1995;7:3–16. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
