A novel approach to measure the contribution of matrix metalloproteinase in the overall net proteolytic activity present in synovial fluids of patients with arthritis
- PMID: 16859524
- PMCID: PMC1779417
- DOI: 10.1186/ar2014
A novel approach to measure the contribution of matrix metalloproteinase in the overall net proteolytic activity present in synovial fluids of patients with arthritis
Abstract
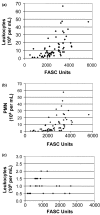
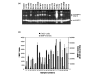
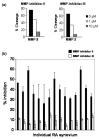
Despite decades of research, only a very limited number of matrix metalloproteinase (MMP) inhibitors have been successful in clinical trials of arthritis. One of the central problems associated with this failure may be our inability to monitor the local activity of proteases in the joints since the integrity of the extracellular matrix results from an equilibrium between noncovalent, 1:1 stoichiometric binding of protease inhibitors to the catalytic site of the activated forms of the enzymes. In the present work, we have measured by flow cytometry the net proteolytic activity in synovial fluids (SF) collected from 95 patients with osteoarthritis and various forms of inflammatory arthritis, including rheumatoid arthritis, spondyloarthropathies, and chronic juvenile arthritis. We found that SF of patients with inflammatory arthritis had significantly higher levels of proteolytic activity than those of osteoarthritis patients. Moreover, the overall activity in inflammatory arthritis patients correlated positively with the number of infiltrated leukocytes and the serum level of C-reactive protein. No such correlations were found in osteoarthritis patients. Members of the MMP family contributed significantly to the proteolytic activity found in SF. Small-molecular-weight MMP inhibitors were indeed effective for inhibiting proteolytic activity in SF, but their effectiveness varied greatly among patients. Interestingly, the contribution of MMPs decreased in patients with very high proteolytic activity, and this was due both to a molar excess of tissue inhibitor of MMP-1 and to an increased contribution of other proteolytic enzymes. These results emphasize the diversity of the MMPs involved in arthritis and, from a clinical perspective, suggest an interesting alternative for testing the potential of new protease inhibitors for the treatment of arthritis.
Figures


Similar articles
-
Levels of matrix metalloproteinase (MMP)-1 in paired sera and synovial fluids of juvenile idiopathic arthritis patients: relationship to inflammatory activity, MMP-3 and tissue inhibitor of metalloproteinases-1 in a longitudinal study.Rheumatology (Oxford). 2005 Nov;44(11):1383-9. doi: 10.1093/rheumatology/kei025. Epub 2005 Jul 27. Rheumatology (Oxford). 2005. PMID: 16049050
-
[Dynamics of matrix metalloproteinase (MMP)-13 in the patients with rheumatoid arthritis].Ryumachi. 2002 Feb;42(1):60-9. Ryumachi. 2002. PMID: 11925908 Japanese.
-
Matrix metalloproteinases MMP-3 and MMP-1 levels in sera and synovial fluids in patients with rheumatoid arthritis and osteoarthritis.Ital J Biochem. 2005 Sep-Dec;54(3-4):248-57. Ital J Biochem. 2005. PMID: 16688934
-
Matrix metalloproteinase expression in the spondyloarthropathies.Curr Opin Rheumatol. 2006 Jul;18(4):364-8. doi: 10.1097/01.bor.0000231904.04548.09. Curr Opin Rheumatol. 2006. PMID: 16763456 Review.
-
Understanding the role of tissue degrading enzymes and their inhibitors in development and disease.Best Pract Res Clin Rheumatol. 2006 Oct;20(5):983-1002. doi: 10.1016/j.berh.2006.06.007. Best Pract Res Clin Rheumatol. 2006. PMID: 16980219 Review.
References
-
- Ahrens D, Koch AE, Pope RM, Stein-Picarella M, Niedbala MJ. Expression of matrix metalloproteinase 9 (96-kd gelatinase B) in human rheumatoid arthritis. Arthritis Rheum. 1996;39:1576–1587. - PubMed
-
- Yoshihara Y, Nakamura H, Obata K, Yamada H, Hayakawa T, Fujikawa K, Okada Y. Matrix metalloproteinases and tissue inhibitors of metalloproteinases in synovial fluids from patients with rheumatoid arthritis or osteoarthritis. Ann Rheum Dis. 2000;59:455–461. doi: 10.1136/ard.59.6.455. - DOI - PMC - PubMed
-
- Smeets TJ, Barg EC, Kraan MC, Smith MD, Breedveld FC, Tak PP. Analysis of the cell infiltrate and expression of proinflammatory cytokines and matrix metalloproteinases in arthroscopic synovial biopsies: comparison with synovial samples from patients with end stage, destructive rheumatoid arthritis. Ann Rheum Dis. 2003;62:635–638. doi: 10.1136/ard.62.7.635. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

