Comparative antibody-mediated phagocytosis of Staphylococcus epidermidis cells grown in a biofilm or in the planktonic state
- PMID: 16861673
- PMCID: PMC1539625
- DOI: 10.1128/IAI.00230-06
Comparative antibody-mediated phagocytosis of Staphylococcus epidermidis cells grown in a biofilm or in the planktonic state
Abstract
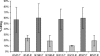
Staphylococcus epidermidis is an important cause of nosocomial infections. Virulence is attributable to elaboration of biofilms on medical surfaces that protect the organisms from immune system clearance. Even though leukocytes can penetrate biofilms, they fail to phagocytose and kill bacteria. The properties that make biofilm bacteria resistant to the immune system are not well characterized. In order to better understand the mechanisms of resistance of bacteria in biofilms to the immune system, we evaluated antibody penetration throughout the biofilm and antibody-mediated phagocytic killing of planktonic versus biofilm cells of S. epidermidis by using a rabbit antibody to poly-N-acetylglucosamine (PNAG). These antibodies are opsonic and protect against infection with planktonic cells of PNAG-positive Staphylococcus aureus and S. epidermidis. Antibody to PNAG readily penetrated the biofilm and bound to the same areas in the biofilm as did wheat germ agglutinin, a lectin known to bind to components of staphylococcal biofilms. However, biofilm cells were more resistant to opsonic killing than their planktonic counterparts in spite of producing more PNAG per cell than planktonic cells. Biofilm extracts inhibited opsonic killing mediated by antibody to PNAG, suggesting that the PNAG antigen within the biofilm matrix prevents antibody binding close to the bacterial cell surface, which is needed for efficient opsonic killing. Increased resistance of biofilm cells to opsonic killing mediated by an otherwise protective antibody was due not to a biofilm-specific phenotype but rather to high levels of antigen within the biofilm that prevented bacterial opsonization by the antibody.
Figures



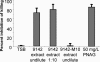
References
-
- Cerca, N., S. Martins, F. Cerca, K. K. Jefferson, G. B. Pier, R. Oliveira, and J. Azeredo. 2005. Comparative assessment of antibiotic susceptibility of coagulase-negative staphylococci in biofilm versus planktonic culture as assessed by bacterial enumeration or rapid XTT colorimetry. J. Antimicrob. Chemother. 56:331-336. - PMC - PubMed
-
- Cerca, N., S. Martins, S. Sillankorva, K. K. Jefferson, G. B. Pier, R. Oliveira, and J. Azeredo. 2005. Effects of growth in the presence of subinhibitory concentrations of dicloxacillin on Staphylococcus epidermidis and Staphylococcus haemolyticus biofilms. Appl. Environ. Microbiol. 71:8677-8682. - PMC - PubMed
-
- Chu, V. H., D. R. Crosslin, J. Y. Friedman, S. D. Reed, C. H. Cabell, R. I. Griffiths, L. E. Masselink, K. S. Kaye, G. R. Corey, L. B. Reller, M. E. Stryjewski, K. A. Schulman, and V. G. Fowler, Jr. 2005. Staphylococcus aureus bacteremia in patients with prosthetic devices: costs and outcomes. Am. J. Med. 118:1416.e19-1416.e22. - PubMed
-
- Costerton, J., P. Stewart, and P. Greenberg. 1999. Bacterial biofilms: a common cause of persistent infections. Science 284:1318-1322. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

