Tyrosine phosphatases epsilon and alpha perform specific and overlapping functions in regulation of voltage-gated potassium channels in Schwann cells
- PMID: 16870705
- PMCID: PMC1635364
- DOI: 10.1091/mbc.e06-02-0151
Tyrosine phosphatases epsilon and alpha perform specific and overlapping functions in regulation of voltage-gated potassium channels in Schwann cells
Abstract
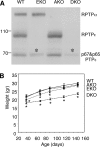
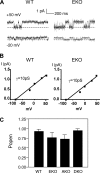
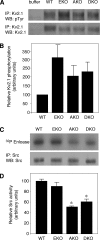
Tyrosine phosphatases (PTPs) epsilon and alpha are closely related and share several molecular functions, such as regulation of Src family kinases and voltage-gated potassium (Kv) channels. Functional interrelationships between PTPepsilon and PTPalpha and the mechanisms by which they regulate K+ channels and Src were analyzed in vivo in mice lacking either or both PTPs. Lack of either PTP increases Kv channel activity and phosphorylation in Schwann cells, indicating these PTPs inhibit Kv current amplitude in vivo. Open probability and unitary conductance of Kv channels are unchanged, suggesting an effect on channel number or organization. PTPalpha inhibits Kv channels more strongly than PTPepsilon; this correlates with constitutive association of PTPalpha with Kv2.1, driven by membranal localization of PTPalpha. PTPalpha, but not PTPepsilon, activates Src in sciatic nerve extracts, suggesting Src deregulation is not responsible exclusively for the observed phenotypes and highlighting an unexpected difference between both PTPs. Developmentally, sciatic nerve myelination is reduced transiently in mice lacking either PTP and more so in mice lacking both PTPs, suggesting both PTPs support myelination but are not fully redundant. We conclude that PTPepsilon and PTPalpha differ significantly in their regulation of Kv channels and Src in the system examined and that similarity between PTPs does not necessarily result in full functional redundancy in vivo.
Figures





Similar articles
-
Hypomyelination and increased activity of voltage-gated K(+) channels in mice lacking protein tyrosine phosphatase epsilon.EMBO J. 2000 Aug 1;19(15):4036-45. doi: 10.1093/emboj/19.15.4036. EMBO J. 2000. PMID: 10921884 Free PMC article.
-
Phosphorylation-dependent regulation of Kv2.1 Channel activity at tyrosine 124 by Src and by protein-tyrosine phosphatase epsilon.J Biol Chem. 2003 May 9;278(19):17509-14. doi: 10.1074/jbc.M212766200. Epub 2003 Mar 1. J Biol Chem. 2003. PMID: 12615930
-
Constitutive activation of delayed-rectifier potassium channels by a src family tyrosine kinase in Schwann cells.EMBO J. 1998 Aug 17;17(16):4723-34. doi: 10.1093/emboj/17.16.4723. EMBO J. 1998. PMID: 9707431 Free PMC article.
-
Protein tyrosine phosphatase alpha (PTPalpha): a Src family kinase activator and mediator of multiple biological effects.Curr Top Med Chem. 2003;3(7):821-35. doi: 10.2174/1568026033452320. Curr Top Med Chem. 2003. PMID: 12678847 Review.
-
Src kinase regulation by phosphorylation and dephosphorylation.Biochem Biophys Res Commun. 2005 May 27;331(1):1-14. doi: 10.1016/j.bbrc.2005.03.012. Biochem Biophys Res Commun. 2005. PMID: 15845350 Review.
Cited by
-
Leptin modulates the intrinsic excitability of AgRP/NPY neurons in the arcuate nucleus of the hypothalamus.J Neurosci. 2014 Apr 16;34(16):5486-96. doi: 10.1523/JNEUROSCI.4861-12.2014. J Neurosci. 2014. PMID: 24741039 Free PMC article.
-
Protein-tyrosine phosphatase alpha regulates stem cell factor-dependent c-Kit activation and migration of mast cells.J Biol Chem. 2008 Oct 24;283(43):29175-85. doi: 10.1074/jbc.M804077200. Epub 2008 Aug 25. J Biol Chem. 2008. PMID: 18725415 Free PMC article.
-
Oxidation of KCNB1 K(+) channels in central nervous system and beyond.World J Biol Chem. 2014 May 26;5(2):85-92. doi: 10.4331/wjbc.v5.i2.85. World J Biol Chem. 2014. PMID: 24921000 Free PMC article. Review.
-
Dynamic modulation of the kv2.1 channel by SRC-dependent tyrosine phosphorylation.J Proteome Res. 2012 Feb 3;11(2):1018-26. doi: 10.1021/pr200770v. Epub 2011 Dec 7. J Proteome Res. 2012. PMID: 22106938 Free PMC article.
-
Role of protein phosphatase 2A in kidney disease (Review).Exp Ther Med. 2021 Nov;22(5):1236. doi: 10.3892/etm.2021.10671. Epub 2021 Aug 31. Exp Ther Med. 2021. PMID: 34539832 Free PMC article. Review.
References
-
- Alonso A., Sasin J., Bottini N., Friedberg I., Osterman A., Godzik A., Hunter T., Dixon J., Mustelin T. Protein tyrosine phosphatases in the human genome. Cell. 2004;117:699–711. - PubMed
-
- Andersen J. N., Jansen P. G., Echwald S. M., Mortensen O. H., Fukada T., Del Vecchio R., Tonks N. K., Moller N. P. A genomic perspective on protein tyrosine phosphatases: gene structure, pseudogenes, and genetic disease linkage. FASEB J. 2004;18:8–30. - PubMed
-
- Bjorge J. D., Pang A., Fujita D. J. Identification of protein-tyrosine phosphatase 1B as the major tyrosine phosphatase activity capable of dephosphorylating and activating c-Src in several human breast cancer cell lines. J. Biol. Chem. 2000;275:41439–41446. - PubMed
-
- Blanchetot C., Chagnon M., Dube N., Halle M., Tremblay M. L. Substrate-trapping techniques in the identification of cellular PTP targets. Methods. 2005;35:44–53. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

