The role of enzyme dynamics and tunnelling in catalysing hydride transfer: studies of distal mutants of dihydrofolate reductase
- PMID: 16873118
- PMCID: PMC1647312
- DOI: 10.1098/rstb.2006.1871
The role of enzyme dynamics and tunnelling in catalysing hydride transfer: studies of distal mutants of dihydrofolate reductase
Abstract
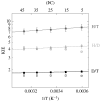
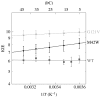
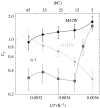
Residues M42 and G121 of Escherichia coli dihydrofolate reductase (ecDHFR) are on opposite sides of the catalytic centre (15 and 19 A away from it, respectively). Theoretical studies have suggested that these distal residues might be part of a dynamics network coupled to the reaction catalysed at the active site. The ecDHFR mutant G121V has been extensively studied and appeared to have a significant effect on rate, but only a mild effect on the nature of H-transfer. The present work examines the effect of M42W on the physical nature of the catalysed hydride transfer step. Intrinsic kinetic isotope effects (KIEs), their temperature dependence and activation parameters were studied. The findings presented here are in accordance with the environmentally coupled hydrogen tunnelling. In contrast to the wild-type (WT), fluctuations of the donor-acceptor distance were required, leading to a significant temperature dependence of KIEs and deflated intercepts. A comparison of M42W and G121V to the WT enzyme revealed that the reduced rates, the inflated primary KIEs and their temperature dependences resulted from an imperfect potential surface pre-arrangement relative to the WT enzyme. Apparently, the coupling of the enzyme's dynamics to the reaction coordinate was altered by the mutation, supporting the models in which dynamics of the whole protein is coupled to its catalysed chemistry.
Figures
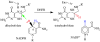
Similar articles
-
Effects of a distal mutation on active site chemistry.Biochemistry. 2006 Feb 7;45(5):1383-92. doi: 10.1021/bi0518242. Biochemistry. 2006. PMID: 16445280 Free PMC article.
-
Coupling of protein motions and hydrogen transfer during catalysis by Escherichia coli dihydrofolate reductase.Biochem J. 2006 Feb 15;394(Pt 1):259-65. doi: 10.1042/BJ20051464. Biochem J. 2006. PMID: 16241906 Free PMC article.
-
Extension and limits of the network of coupled motions correlated to hydride transfer in dihydrofolate reductase.J Am Chem Soc. 2014 Feb 12;136(6):2575-82. doi: 10.1021/ja411998h. Epub 2014 Jan 31. J Am Chem Soc. 2014. PMID: 24450297 Free PMC article.
-
Multiple intermediates, diverse conformations, and cooperative conformational changes underlie the catalytic hydride transfer reaction of dihydrofolate reductase.Top Curr Chem. 2013;337:165-87. doi: 10.1007/128_2012_408. Top Curr Chem. 2013. PMID: 23420416 Free PMC article. Review.
-
Insights into enzyme function from studies on mutants of dihydrofolate reductase.Science. 1988 Mar 4;239(4844):1105-10. doi: 10.1126/science.3125607. Science. 1988. PMID: 3125607 Review.
Cited by
-
Relationship of femtosecond-picosecond dynamics to enzyme-catalyzed H-transfer.Top Curr Chem. 2013;337:1-39. doi: 10.1007/128_2012_407. Top Curr Chem. 2013. PMID: 23539379 Free PMC article. Review.
-
Examinations of the Chemical Step in Enzyme Catalysis.Methods Enzymol. 2016;577:287-318. doi: 10.1016/bs.mie.2016.05.017. Epub 2016 Jun 28. Methods Enzymol. 2016. PMID: 27498642 Free PMC article.
-
Tuning of the H-transfer coordinate in primitive versus well-evolved enzymes.Chemphyschem. 2008 May 16;9(7):980-2. doi: 10.1002/cphc.200800067. Chemphyschem. 2008. PMID: 18444258 Free PMC article. No abstract available.
-
Hydrogen tunneling links protein dynamics to enzyme catalysis.Annu Rev Biochem. 2013;82:471-96. doi: 10.1146/annurev-biochem-051710-133623. Annu Rev Biochem. 2013. PMID: 23746260 Free PMC article. Review.
-
Coordinated effects of distal mutations on environmentally coupled tunneling in dihydrofolate reductase.Proc Natl Acad Sci U S A. 2006 Oct 24;103(43):15753-8. doi: 10.1073/pnas.0606976103. Epub 2006 Oct 10. Proc Natl Acad Sci U S A. 2006. PMID: 17032759 Free PMC article.
References
-
- Agarwal P.K, Billeter S.R, Hammes-Schiffer S. Nuclear quantum effects and enzyme dynamics in dihydrofolate reductase catalysis. J. Phys. Chem. B. 2002a;106:3283–3293. doi: 10.1021/jp020190v. - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

