Continued administration of ciliary neurotrophic factor protects mice from inflammatory pathology in experimental autoimmune encephalomyelitis
- PMID: 16877358
- PMCID: PMC1698786
- DOI: 10.2353/ajpath.2006.051086
Continued administration of ciliary neurotrophic factor protects mice from inflammatory pathology in experimental autoimmune encephalomyelitis
Abstract
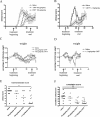
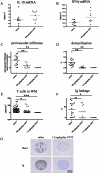
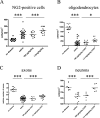
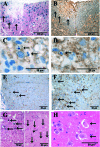
Multiple sclerosis is an inflammatory disease of the central nervous system that leads to loss of myelin and oligodendrocytes and damage to axons. We show that daily administration (days 8 to 24) of murine ciliary neurotrophic factor (CNTF), a neurotrophic factor that has been described as a survival and differentiation factor for neurons and oligodendrocytes, significantly ameliorates the clinical course of a mouse model of multiple sclerosis. In the acute phase of experimental autoimmune encephalomyelitis induced by myelin oligodendrocyte glycoprotein peptide 35-55, treatment with CNTF did not change the peripheral immune response but did reduce the number of perivascular infiltrates and T cells and the level of diffuse microglial activation in spinal cord. Blood brain barrier permeability was significantly reduced in CNTF-treated animals. Beneficial effects of CNTF did not persist after it was withdrawn. After cessation of CNTF treatment, inflammation and symptoms returned to control levels. However, slight but significantly higher numbers of oligodendrocytes, NG2-positive cells, axons, and neurons were observed in mice that had been treated with high concentrations of CNTF. Our results show that CNTF inhibits inflammation in the spinal cord, resulting in amelioration of the clinical course of experimental autoimmune encephalomyelitis during time of treatment.
Figures

Similar articles
-
Phosphodiesterase 5 inhibition at disease onset prevents experimental autoimmune encephalomyelitis progression through immunoregulatory and neuroprotective actions.Exp Neurol. 2014 Jan;251:58-71. doi: 10.1016/j.expneurol.2013.10.021. Epub 2013 Nov 7. Exp Neurol. 2014. PMID: 24211383
-
Endogenous leukemia inhibitory factor production limits autoimmune demyelination and oligodendrocyte loss.Glia. 2006 May;53(7):696-703. doi: 10.1002/glia.20321. Glia. 2006. PMID: 16498619
-
Overexpression of CNTF in Mesenchymal Stem Cells reduces demyelination and induces clinical recovery in experimental autoimmune encephalomyelitis mice.J Neuroimmunol. 2009 Jan 3;206(1-2):58-69. doi: 10.1016/j.jneuroim.2008.10.014. Epub 2008 Dec 9. J Neuroimmunol. 2009. PMID: 19081144
-
Function of neurotrophic factors beyond the nervous system: inflammation and autoimmune demyelination.Crit Rev Immunol. 2009;29(1):43-68. doi: 10.1615/critrevimmunol.v29.i1.20. Crit Rev Immunol. 2009. PMID: 19348610 Review.
-
Growth factors and myelin regeneration in multiple sclerosis.Mult Scler. 1997 Apr;3(2):113-20. doi: 10.1177/135245859700300210. Mult Scler. 1997. PMID: 9291164 Review.
Cited by
-
A granulocyte-macrophage colony-stimulating factor and interleukin-15 fusokine induces a regulatory B cell population with immune suppressive properties.Nat Med. 2009 Sep;15(9):1038-45. doi: 10.1038/nm.2003. Epub 2009 Aug 9. Nat Med. 2009. PMID: 19668193
-
IL-11 regulates autoimmune demyelination.J Immunol. 2009 Oct 1;183(7):4229-40. doi: 10.4049/jimmunol.0900622. Epub 2009 Sep 4. J Immunol. 2009. PMID: 19734214 Free PMC article.
-
AMP-activated protein kinase signaling protects oligodendrocytes that restore central nervous system functions in an experimental autoimmune encephalomyelitis model.Am J Pathol. 2013 Aug;183(2):526-41. doi: 10.1016/j.ajpath.2013.04.030. Epub 2013 Jun 10. Am J Pathol. 2013. PMID: 23759513 Free PMC article.
-
Lentivirus-mediated Persephin over-expression in Parkinson's disease rats.Neural Regen Res. 2015 Nov;10(11):1814-8. doi: 10.4103/1673-5374.170309. Neural Regen Res. 2015. PMID: 26807117 Free PMC article.
-
Promoting myelin repair and return of function in multiple sclerosis.FEBS Lett. 2011 Dec 1;585(23):3813-20. doi: 10.1016/j.febslet.2011.08.017. Epub 2011 Aug 18. FEBS Lett. 2011. PMID: 21864535 Free PMC article. Review.
References
-
- Keegan BM, Noseworthy JH. Multiple sclerosis. Annu Rev Med. 2002;53:285–302. - PubMed
-
- Owens T. The enigma of multiple sclerosis. Curr Opin Neurol. 2003;16:259–265. - PubMed
-
- Prineas JW. The neuropathology of multiple sclerosis. Koetsier JC, editor. Elsevier Science Publishers,; Amsterdam: Demyelinating Diseases. 1985:pp 213–257.
-
- Lassmann H. Springer-Verlag,; Berlin: Comparative Neuropathology of Chronic Experimental Allergic Encephalomyelitis and Multiple Sclerosis. 1983 - PubMed
-
- Bjartmar C, Kidd GJ, Mork S, Rudick R, Trapp BD. Neurological disability correlates with spinal cord axonal loss and reduced N-acetyl aspartate in chronic multiple sclerosis patients. Ann Neurol. 2000;48:893–901. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

