Lens-specific expression of TGF-beta induces anterior subcapsular cataract formation in the absence of Smad3
- PMID: 16877415
- PMCID: PMC2811063
- DOI: 10.1167/iovs.05-1208
Lens-specific expression of TGF-beta induces anterior subcapsular cataract formation in the absence of Smad3
Abstract
Purpose: Smad3, a mediator of TGF-beta signaling has been shown to be involved in the epithelial-to-mesenchymal transformation (EMT) of lens epithelial cells in a lens injury model. In this study, the role of Smad3 in anterior subcapsular cataract (ASC) formation was investigated in a transgenic TGF-beta/Smad3 knockout mouse model.
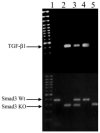
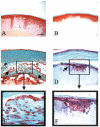
Methods: TGF-beta1 transgenic mice (containing a human TGF-beta1 cDNA construct expressed under the alphaA-crystallin promoter) were bred with mice on a Smad3-null background to generate mice with the following genotypes: TGF-beta1/Smad3(-/-) (null), TGF-beta1/Smad3(+/-), TGF-beta1/Smad3(+/+), and nontransgenic/Smad3(+/+). Lenses from mice of each genotype were dissected and prepared for histologic or optical analyses.
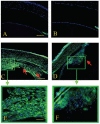
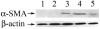
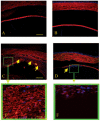
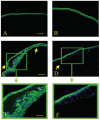
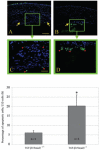
Results: All transgenic TGF-beta1 lenses demonstrated subcapsular plaque formation and EMT as indicated by the expression of alpha-smooth muscle actin. However, the sizes of the plaques were reduced in the TGF-beta1/Smad3(-/-) lenses, as was the level of type IV collagen deposition when compared with TGF-beta1/Smad3(+/-) and TGF-beta1/Smad3(+/+) lenses. An increased number of apoptotic figures was also observed in the plaques of the TGF-beta1/Smad3(-/-) lenses compared with TGF-beta1/Smad3(+/+) littermates.
Conclusions: Lens-specific expression of TGF-beta1 induced ASC formation in the absence of the Smad3 signaling mediator, suggests that alternative TGF-beta-signaling pathways participate in this ocular fibrotic model.
Figures
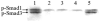





References
-
- Qing J, Zhang Y, Derynck R. Structural and functional characterization of the transforming growth factor-beta-induced Smad3/c-Jun transcriptional cooperativity. J Biol Chem. 2000;275:38802–38812. - PubMed
-
- Dunker N, Krieglstein K. Reduced programmed cell death in the retina and defects in lens and cornea of Tgfbeta2(-/-) Tgf-beta3(-/-) double-deficient mice. Cell Tissue Res. 2003;313:1–10. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

