Antisense oligonucleotide therapy for neurodegenerative disease
- PMID: 16878173
- PMCID: PMC1518790
- DOI: 10.1172/JCI25424
Antisense oligonucleotide therapy for neurodegenerative disease
Abstract
Neurotoxicity from accumulation of misfolded/mutant proteins is thought to drive pathogenesis in neurodegenerative diseases. Since decreasing levels of proteins responsible for such accumulations is likely to ameliorate disease, a therapeutic strategy has been developed to downregulate almost any gene in the CNS. Modified antisense oligonucleotides, continuously infused intraventricularly, have been demonstrated to distribute widely throughout the CNS of rodents and primates, including the regions affected in the major neurodegenerative diseases. Using this route of administration, we found that antisense oligonucleotides to superoxide dismutase 1 (SOD1), one of the most abundant brain proteins, reduced both SOD1 protein and mRNA levels throughout the brain and spinal cord. Treatment initiated near onset significantly slowed disease progression in a model of amyotrophic lateral sclerosis (ALS) caused by a mutation in SOD1. This suggests that direct delivery of antisense oligonucleotides could be an effective, dosage-regulatable means of treating neurodegenerative diseases, including ALS, where appropriate target proteins are known.
Figures
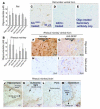


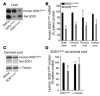
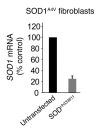

References
-
- Bossy-Wetzel E., Schwarzenbacher R., Lipton S.A. Molecular pathways to neurodegeneration. Nat. Med. 2004;10(Suppl.):S2–S9. - PubMed
-
- Xia H., Mao Q., Paulson H.L., Davidson B.L. siRNA-mediated gene silencing in vitro and in vivo. Nat. Biotechnol. 2002;20:1006–1010. - PubMed
-
- Xia H., et al. RNAi suppresses polyglutamine-induced neurodegeneration in a model of spinocerebellar ataxia. Nat. Med. 2004;10:816–820. - PubMed
-
- Singer O., et al. Targeting BACE1 with siRNAs ameliorates Alzheimer disease neuropathology in a transgenic model. Nat Neurosci. 2005;8:1343–1349. - PubMed
-
- Raoul C., et al. Lentiviral-mediated silencing of SOD1 through RNA interference retards disease onset and progression in a mouse model of ALS. Nat. Med. 2005;11:423–428. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

