Structural and biochemical characterization of a mitochondrial peroxiredoxin from Plasmodium falciparum
- PMID: 16879648
- PMCID: PMC1618809
- DOI: 10.1111/j.1365-2958.2006.05303.x
Structural and biochemical characterization of a mitochondrial peroxiredoxin from Plasmodium falciparum
Abstract
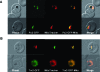
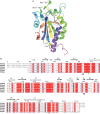
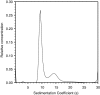
Plasmodium falciparum possesses a single mitochondrion with a functional electron transport chain. During respiration, reactive oxygen species are generated that need to be removed to protect the organelle from oxidative damage. In the absence of catalase and glutathione peroxidase, the parasites rely primarily on peroxiredoxin-linked systems for protection. We have analysed the biochemical and structural features of the mitochondrial peroxiredoxin and thioredoxin of P. falciparum. The mitochondrial localization of both proteins was confirmed by expressing green fluorescent protein fusions in parasite erythrocytic stages. Recombinant protein was kinetically characterized using the cytosolic and the mitochondrial thioredoxin (PfTrx1 and PfTrx2 respectively). The peroxiredoxin clearly preferred PfTrx2 to PfTrx1 as a reducing partner, reflected by the KM values of 11.6 microM and 130.4 microM respectively. Substitution of the two dyads asparagine-62/tyrosine-63 and phenylalanine-139/alanine-140 residues by aspartate-phenylalaine and valine-serine, respectively, reduced the KM for Trx1 but had no effect on the KM of Trx2 suggesting some role for these residues in the discrimination between the two substrates. Solution studies suggest that the protein exists primarily in a homodecameric form. The crystal structure of the mitochondrial peroxiredoxin reveals a fold typical of the 2-Cys class peroxiredoxins and a dimeric form with an intermolecular disulphide bridge between Cys67 and Cys187. These results show that the mitochondrial peroxiredoxin of P. falciparum occurs in both dimeric and decameric forms when purified under non-reducing conditions.
Figures

Similar articles
-
Thioredoxin networks in the malarial parasite Plasmodium falciparum.Antioxid Redox Signal. 2006 Jul-Aug;8(7-8):1227-39. doi: 10.1089/ars.2006.8.1227. Antioxid Redox Signal. 2006. PMID: 16910770 Review.
-
Plasmodium falciparum 2-Cys peroxiredoxin reacts with plasmoredoxin and peroxynitrite.Biol Chem. 2005 Nov;386(11):1129-36. doi: 10.1515/BC.2005.129. Biol Chem. 2005. PMID: 16307478
-
2-Cys peroxiredoxin PfTrx-Px1 is involved in the antioxidant defence of Plasmodium falciparum.Mol Biochem Parasitol. 2003 Aug 31;130(2):75-81. doi: 10.1016/s0166-6851(03)00161-0. Mol Biochem Parasitol. 2003. PMID: 12946843
-
Redox and antioxidant systems of the malaria parasite Plasmodium falciparum.Mol Microbiol. 2004 Sep;53(5):1291-305. doi: 10.1111/j.1365-2958.2004.04257.x. Mol Microbiol. 2004. PMID: 15387810 Review.
-
Crystal structure of a novel Plasmodium falciparum 1-Cys peroxiredoxin.J Mol Biol. 2005 Mar 4;346(4):1021-34. doi: 10.1016/j.jmb.2004.12.022. Epub 2005 Jan 8. J Mol Biol. 2005. PMID: 15701514
Cited by
-
Dual targeting of antioxidant and metabolic enzymes to the mitochondrion and the apicoplast of Toxoplasma gondii.PLoS Pathog. 2007 Aug 31;3(8):e115. doi: 10.1371/journal.ppat.0030115. PLoS Pathog. 2007. PMID: 17784785 Free PMC article.
-
Structural insights into thioredoxin-2: a component of malaria parasite protein secretion machinery.Sci Rep. 2011;1:179. doi: 10.1038/srep00179. Epub 2011 Dec 1. Sci Rep. 2011. PMID: 22355694 Free PMC article.
-
How pH modulates the dimer-decamer interconversion of 2-Cys peroxiredoxins from the Prx1 subfamily.J Biol Chem. 2015 Mar 27;290(13):8582-90. doi: 10.1074/jbc.M114.619205. Epub 2015 Feb 9. J Biol Chem. 2015. PMID: 25666622 Free PMC article.
-
Observed octameric assembly of a Plasmodium yoelii peroxiredoxin can be explained by the replacement of native "ball-and-socket" interacting residues by an affinity tag.Protein Sci. 2013 Oct;22(10):1445-52. doi: 10.1002/pro.2328. Protein Sci. 2013. PMID: 23934758 Free PMC article.
-
Structure-based insights into the catalytic power and conformational dexterity of peroxiredoxins.Antioxid Redox Signal. 2011 Aug 1;15(3):795-815. doi: 10.1089/ars.2010.3624. Epub 2011 Apr 20. Antioxid Redox Signal. 2011. PMID: 20969484 Free PMC article. Review.
References
-
- Akerman SE, Müller S. 2-Cys peroxiredoxin PfTrx-Px1 is involved in the antioxidant defence of Plasmodium falciparum. Mol Biochem Parasitol. 2003;130:75–81. - PubMed
-
- Akerman SE, Müller S. Peroxiredoxin-linked detoxification of hydroperoxides in Toxoplasma gondii. J Biol Chem. 2005;280:564–570. - PubMed
-
- Alphey MS, Bond CS, Tetaud E, Fairlamb AH, Hunter WN. The structure of reduced tryparedoxin peroxidase reveals a decamer and insight into reactivity of 2Cys-peroxiredoxins. J Mol Biol. 2000;300:903–916. - PubMed
-
- Argos P. An investigation of protein subunit and domain interfaces. Protein Eng. 1988;2:101–113. - PubMed
-
- Bauer H, Kanzok SM, Schirmer RH. Thioredoxin-2 but not thioredoxin-1 is a substrate of thioredoxin peroxidase-1 from Drosophila melanogaster: isolation and characterization of a second thioredoxin in D. melanogaster and evidence for distinct biological functions of Trx-1 and Trx-2. J Biol Chem. 2002;277:17457–17463. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

