Tetanus toxin is internalized by a sequential clathrin-dependent mechanism initiated within lipid microdomains and independent of epsin1
- PMID: 16880274
- PMCID: PMC2064241
- DOI: 10.1083/jcb.200508170
Tetanus toxin is internalized by a sequential clathrin-dependent mechanism initiated within lipid microdomains and independent of epsin1
Abstract
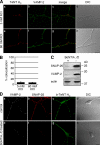
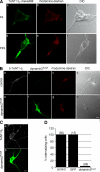
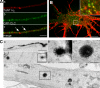
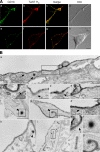
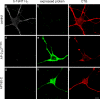
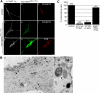
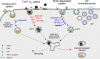
Ligand-receptor complexes are internalized by a variety of endocytic mechanisms. Some are initiated within clathrin-coated membranes, whereas others involve lipid microdomains of the plasma membrane. In neurons, where alternative targeting to short- or long-range trafficking routes underpins the differential processing of synaptic vesicle components and neurotrophin receptors, the mechanism giving access to the axonal retrograde pathway remains unknown. To investigate this sorting process, we examined the internalization of a tetanus neurotoxin fragment (TeNT HC), which shares axonal carriers with neurotrophins and their receptors. Previous studies have shown that the TeNT HC receptor, which comprises polysialogangliosides, resides in lipid microdomains. We demonstrate that TeNT HC internalization also relies on a specialized clathrin-mediated pathway, which is independent of synaptic vesicle recycling. Moreover, unlike transferrin uptake, this AP-2-dependent process is independent of epsin1. These findings identify a pathway for TeNT, beginning with the binding to a lipid raft component (GD1b) and followed by dissociation from GD1b as the toxin internalizes via a clathrin-mediated mechanism using a specific subset of adaptor proteins.
Figures

Similar articles
-
Internalization of a GFP-tetanus toxin C-terminal fragment fusion protein at mature mouse neuromuscular junctions.Mol Cell Neurosci. 2005 Dec;30(4):572-82. Mol Cell Neurosci. 2005. PMID: 16456925
-
Analysis of retrograde transport in motor neurons reveals common endocytic carriers for tetanus toxin and neurotrophin receptor p75NTR.J Cell Biol. 2002 Jan 21;156(2):233-9. doi: 10.1083/jcb.200106142. Epub 2002 Jan 21. J Cell Biol. 2002. PMID: 11807088 Free PMC article.
-
Anthrax toxin triggers endocytosis of its receptor via a lipid raft-mediated clathrin-dependent process.J Cell Biol. 2003 Feb 3;160(3):321-8. doi: 10.1083/jcb.200211018. Epub 2003 Jan 27. J Cell Biol. 2003. PMID: 12551953 Free PMC article.
-
The travel diaries of tetanus and botulinum neurotoxins.Toxicon. 2018 Jun 1;147:58-67. doi: 10.1016/j.toxicon.2017.10.008. Epub 2017 Oct 12. Toxicon. 2018. PMID: 29031941 Review.
-
Cargo recognition during clathrin-mediated endocytosis: a team effort.Curr Opin Cell Biol. 2004 Aug;16(4):392-9. doi: 10.1016/j.ceb.2004.06.001. Curr Opin Cell Biol. 2004. PMID: 15261671 Review.
Cited by
-
A Heterologous Reporter Defines the Role of the Tetanus Toxin Interchain Disulfide in Light-Chain Translocation.Infect Immun. 2015 Jul;83(7):2714-24. doi: 10.1128/IAI.00477-15. Epub 2015 Apr 20. Infect Immun. 2015. PMID: 25895970 Free PMC article.
-
A New Membrane Lipid Raft Gene SpFLT-1 Facilitating the Endocytosis of Vibrio alginolyticus in the Crab Scylla paramamosain.PLoS One. 2015 Jul 17;10(7):e0133443. doi: 10.1371/journal.pone.0133443. eCollection 2015. PLoS One. 2015. PMID: 26186350 Free PMC article.
-
Fragment C of tetanus toxin: new insights into its neuronal signaling pathway.Int J Mol Sci. 2012;13(6):6883-6901. doi: 10.3390/ijms13066883. Epub 2012 Jun 7. Int J Mol Sci. 2012. PMID: 22837670 Free PMC article. Review.
-
Diversity of raft-like domains in late endosomes.PLoS One. 2007 Apr 25;2(4):e391. doi: 10.1371/journal.pone.0000391. PLoS One. 2007. PMID: 17460758 Free PMC article.
-
Specific retrograde transduction of spinal motor neurons using lentiviral vectors targeted to presynaptic NMJ receptors.Mol Ther. 2014 Jul;22(7):1285-1298. doi: 10.1038/mt.2014.49. Epub 2014 Mar 26. Mol Ther. 2014. PMID: 24670531 Free PMC article.
References
-
- Aquino, D.A., M.A. Bisby, and R.W. Ledeen. 1985. Retrograde axonal transport of gangliosides and glycoproteins in the motoneurons of rat sciatic nerve. J. Neurochem. 45:1262–1267. - PubMed
-
- Blanpied, T.A., D.B. Scott, and M.D. Ehlers. 2002. Dynamics and regulation of clathrin coats at specialized endocytic zones of dendrites and spines. Neuron. 36:435–449. - PubMed
-
- Boffey, J., M. Odaka, D. Nicoll, E.R. Wagner, K. Townson, T. Bowes, J. Conner, K. Furukawa, and H.J. Willison. 2005. Characterisation of the immunoglobulin variable region gene usage encoding the murine anti-ganglioside antibody repertoire. J. Neuroimmunol. 165:92–103. - PubMed
-
- Bohnert, S., and G. Schiavo. 2005. Tetanus toxin is transported in a novel neuronal compartment characterized by a specialized pH regulation. J. Biol. Chem. 280:42336–42344. - PubMed

