Prefibrillar amyloid aggregates could be generic toxins in higher organisms
- PMID: 16885229
- PMCID: PMC6673784
- DOI: 10.1523/JNEUROSCI.4809-05.2006
Prefibrillar amyloid aggregates could be generic toxins in higher organisms
Abstract
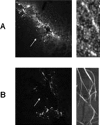
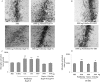
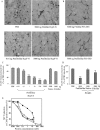
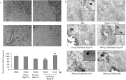
More than 40 human diseases are associated with fibrillar deposits of specific peptides or proteins in tissue. Amyloid fibrils, or their precursors, can be highly toxic to cells, suggesting their key role in disease pathogenesis. Proteins not associated with any disease are able to form oligomers and amyloid assemblies in vitro displaying structures and cytotoxicity comparable with those of aggregates of disease-related polypeptides. In isolated cells, such toxicity has been shown to result from increased membrane permeability with disruption of ion homeostasis and oxidative stress. Here we microinjected into the nucleus basalis magnocellularis of rat brains aggregates of an Src homology 3 domain and the N-terminal domain of the prokaryotic HypF, neither of which is associated with amyloid disease. Prefibrillar aggregates of both proteins, but not their mature fibrils or soluble monomers, impaired cholinergic neuron viability in a dose-dependent manner similar to that seen in cell cultures. Contrary to the situation with cultured cells, however, under our experimental conditions, cell stress in tissue is not followed by a comparable level of cell death, a result that is very likely to reflect the presence of protective mechanisms reducing aggregate toxicity. These findings support the hypothesis that neurodegenerative disorders result primarily from a generic cell dysfunction caused by early misfolded species in the aggregation process.
Figures

Similar articles
-
Glycosaminoglycans (GAGs) suppress the toxicity of HypF-N prefibrillar aggregates.J Mol Biol. 2012 Aug 24;421(4-5):616-30. doi: 10.1016/j.jmb.2012.02.007. Epub 2012 Feb 9. J Mol Biol. 2012. PMID: 22326346
-
Proteomic analysis of cells exposed to prefibrillar aggregates of HypF-N.Biochim Biophys Acta. 2009 Aug;1794(8):1243-50. doi: 10.1016/j.bbapap.2009.04.009. Epub 2009 May 3. Biochim Biophys Acta. 2009. PMID: 19409514
-
Transthyretin suppresses the toxicity of oligomers formed by misfolded proteins in vitro.Biochim Biophys Acta. 2013 Dec;1832(12):2302-14. doi: 10.1016/j.bbadis.2013.09.011. Epub 2013 Sep 25. Biochim Biophys Acta. 2013. PMID: 24075940
-
Soluble protein oligomers as emerging toxins in Alzheimer's and other amyloid diseases.IUBMB Life. 2007 Apr-May;59(4-5):332-45. doi: 10.1080/15216540701283882. IUBMB Life. 2007. PMID: 17505973 Review.
-
Structure of amyloid oligomers and their mechanisms of toxicities: Targeting amyloid oligomers using novel therapeutic approaches.Eur J Med Chem. 2016 May 23;114:41-58. doi: 10.1016/j.ejmech.2016.02.065. Epub 2016 Mar 2. Eur J Med Chem. 2016. PMID: 26974374 Review.
Cited by
-
Aggregation of Aß(25-35) on DOPC and DOPC/DHA bilayers: an atomic force microscopy study.PLoS One. 2014 Dec 31;9(12):e115780. doi: 10.1371/journal.pone.0115780. eCollection 2014. PLoS One. 2014. PMID: 25551704 Free PMC article.
-
Cooperative amyloid fibre binding and disassembly by the Hsp70 disaggregase.EMBO J. 2022 Aug 16;41(16):e110410. doi: 10.15252/embj.2021110410. Epub 2022 Jun 13. EMBO J. 2022. PMID: 35698800 Free PMC article.
-
Screening for toxic amyloid in yeast exemplifies the role of alternative pathway responsible for cytotoxicity.PLoS One. 2009;4(3):e4539. doi: 10.1371/journal.pone.0004539. Epub 2009 Mar 5. PLoS One. 2009. PMID: 19262694 Free PMC article.
-
Structural classification of toxic amyloid oligomers.J Biol Chem. 2008 Oct 31;283(44):29639-43. doi: 10.1074/jbc.R800016200. Epub 2008 Aug 22. J Biol Chem. 2008. PMID: 18723507 Free PMC article. Review.
-
Protein fibrillation lag times during kinetic inhibition.Biophys J. 2014 Aug 5;107(3):711-720. doi: 10.1016/j.bpj.2014.06.029. Biophys J. 2014. PMID: 25099810 Free PMC article.
References
-
- Brofman FC, Moechars D, Van Leuven F (2000). Acetylcholinesterase-positive fiber deafferentation and cell shrinkage in the septohippocampal pathway of aged amyloid precursor protein London mutant transgenic mice. Neurobiol Dis 7:152–168. - PubMed
-
- Bucciantini M, Giannoni E, Chiti F, Baroni F, Formigli L, Zurdo J, Taddei N, Ramponi G, Dobson CM, Stefani M (2002). Inherent toxicity of aggregates implies a common mechanism for protein misfolding diseases. Nature 416:507–511. - PubMed
-
- Bucciantini M, Calloni G, Chiti F, Formigli L, Nosi D, Dobson CM, Stefani M (2004). Pre-fibrillar amyloid protein aggregates share common features of cytotoxicity. J Biol Chem 279:31374–31382. - PubMed
-
- Bucciantini M, Rigacci S, Berti A, Pieri L, Cecchi C, Nosi D, Formigli L, Chiti F, Stefani M (2005). Patterns of cell death triggered in two different cell lines by HypF-N prefibrillar aggregates. FASEB J 19:437–439. - PubMed
-
- Carulla N, Caddy GL, Hall DR, Zurdo J, Gairi M, Felix M, Giralt E, Robinson CV, Dobson CM (2005). Molecular recycling within amyloid fibrils. Nature 436:584–588. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous
