TOR regulates late steps of ribosome maturation in the nucleoplasm via Nog1 in response to nutrients
- PMID: 16888624
- PMCID: PMC1553199
- DOI: 10.1038/sj.emboj.7601262
TOR regulates late steps of ribosome maturation in the nucleoplasm via Nog1 in response to nutrients
Abstract
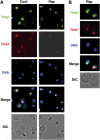
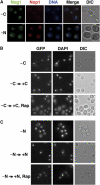
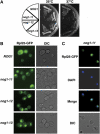
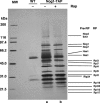
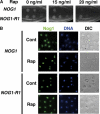
The protein kinase TOR (target of rapamycin) controls several steps of ribosome biogenesis, including gene expression of rRNA and ribosomal proteins, and processing of the 35S rRNA precursor, in the budding yeast Saccharomyces cerevisiae. Here we show that TOR also regulates late stages of ribosome maturation in the nucleoplasm via the nuclear GTP-binding protein Nog1. Nog1 formed a complex that included 60S ribosomal proteins and pre-ribosomal proteins Nop7 and Rlp24. The Nog1 complex shuttled between the nucleolus and the nucleoplasm for ribosome biogenesis, but it was tethered to the nucleolus by both nutrient depletion and TOR inactivation, causing cessation of the late stages of ribosome biogenesis. Furthermore, after this, Nog1 and Nop7 proteins were lost, leading to complete cessation of ribosome maturation. Thus, the Nog1 complex is a critical regulator of ribosome biogenesis mediated by TOR. This is the first description of a physiological regulation of nucleolus-to-nucleoplasm translocation of pre-ribosome complexes.
Figures




References
-
- Allen NP, Huang L, Burlingame A, Rexach M (2001) Proteomic analysis of nucleoporin interacting proteins. J Biol Chem 276: 29268–29274 - PubMed
-
- Azzam R, Chen SL, Shou W, Mah AS, Alexandru G, Nasmyth K, Annan RS, Carr SA, Deshaies RJ (2004) Phosphorylation by cyclin B–Cdk underlies release of mitotic exit activator Cdc14 from the nucleolus. Science 305: 516–519 - PubMed
-
- Bassler J, Grandi P, Gadal O, Lessmann T, Petfalski E, Tollervey D, Lechner J, Hurt E (2001) Identification of a 60S preribosomal particle that is closely linked to nuclear export. Mol Cell 8: 517–529 - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

