Analysis of mitogen-activated protein kinase signaling specificity in response to hyperosmotic stress: use of an analog-sensitive HOG1 allele
- PMID: 16896207
- PMCID: PMC1539154
- DOI: 10.1128/EC.00037-06
Analysis of mitogen-activated protein kinase signaling specificity in response to hyperosmotic stress: use of an analog-sensitive HOG1 allele
Abstract
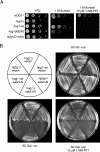
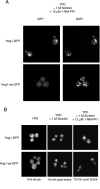
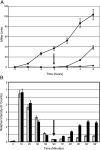
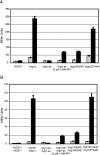
When confronted with a marked increase in external osmolarity, budding yeast (Saccharomyces cerevisiae) cells utilize a conserved mitogen-activated protein kinase (MAPK) signaling cascade (the high-osmolarity glycerol or HOG pathway) to elicit cellular responses necessary to permit continued growth. One input that stimulates the HOG pathway requires the integral membrane protein and putative osmosensor Sho1, which recruits and enables activation of the MAPK kinase kinase Ste11. In mutants that lack the downstream MAPK kinase (pbs2Delta) or the MAPK (hog1Delta) of the HOG pathway, Ste11 activated by hyperosmotic stress is able to inappropriately stimulate the pheromone response pathway. This loss of signaling specificity is known as cross talk. To determine whether it is the Hog1 polypeptide per se or its kinase activity that is necessary to prevent cross talk, we constructed a fully functional analog-sensitive allele of HOG1 to permit acute inhibition of this enzyme without other detectable perturbations of the cell. We found that the catalytic activity of Hog1 is required continuously to prevent cross talk between the HOG pathway and both the pheromone response and invasive growth pathways. Moreover, contrary to previous reports, we found that the kinase activity of Hog1 is necessary for its stress-induced nuclear import. Finally, our results demonstrate a role for active Hog1 in maintaining signaling specificity under conditions of persistently high external osmolarity.
Figures





References
-
- Albertyn, J., S. Hohmann, J. M. Thevelein, and B. A. Prior. 1994. GPD1, which encodes glycerol-3-phosphate dehydrogenase, is essential for growth under osmotic stress in Saccharomyces cerevisiae, and its expression is regulated by the high-osmolarity glycerol response pathway. Mol. Cell. Biol. 14:4135-4144. - PMC - PubMed
-
- Bao, M. Z., M. A. Schwartz, G. T. Cantin, J. R. Yates III, and H. D. Madhani. 2004. Pheromone-dependent destruction of the Tec1 transcription factor is required for MAP kinase signaling specificity in yeast. Cell 119:991-1000. - PubMed
-
- Bardwell, L., J. G. Cook, E. C. Chang, B. R. Cairns, and J. Thorner. 1996. Signaling in the yeast pheromone response pathway: specific and high-affinity interaction of the mitogen-activated protein (MAP) kinases Kss1 and Fus3 with the upstream MAP kinase kinase Ste7. Mol. Cell. Biol. 16:3637-3650. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

