Molecular expression and functional involvement of the bovine calcium-activated chloride channel 1 (bCLCA1) in apical HCO3- permeability of bovine corneal endothelium
- PMID: 16899243
- PMCID: PMC3108034
- DOI: 10.1016/j.exer.2006.06.011
Molecular expression and functional involvement of the bovine calcium-activated chloride channel 1 (bCLCA1) in apical HCO3- permeability of bovine corneal endothelium
Abstract
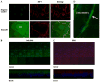
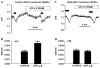
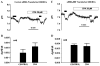
Corneal endothelium secretes HCO(3)(-) from basolateral (stroma) to apical (anterior chamber) compartments. Apical HCO(3)(-) permeability can be enhanced by increasing [Ca(2+)](i). We hypothesized that the bovine calcium-activated chloride channel 1 (bCLCA1), shown previously by PCR screening to be expressed in corneal endothelium, is involved in Ca(2+) activated apical HCO(3)(-) permeability. bCLCA1 expression in cultured bovine corneal endothelial cells (CBCEC) was examined by in situ hybridization analysis, immunoblotting, immunofluorescence and confocal microscopy. Rabbit polyclonal antibodies were generated using a 14 aa polypeptide (417-430) from the predicted sequence of bCLCA1. The small interference RNA (siRNA) knock down technique was used to evaluate the functional involvement of bCLCA1 in apical HCO(3)(-) permeability. In situ hybridization confirmed prominent bCLCA1-specific mRNA expression in CBCEC. bCLCA1 antiserum detected the heterologously expressed bCLCA1 in HEK293 cells and a 90kDa band in CBCEC, which was absent when using the pre-immune serum or antigen absorption of serum. Immunofluoresence staining with anti-bCLCA1 antibody and confocal microscopy indicates an apical membrane location in CBCEC. In CBCEC transfected with bCLCA1 specific siRNA, bCLCA1 expression was reduced by 80%, while transfection with siControl scrambled sequence had no effect. Increasing [Ca(i)(2+)] by application of ATPgammaS or cyclopiazonic acid (CPA) increased apical HCO(3)(-) permeability in siControl transfected CBCEC, while having no effect on apical HCO(3)(-) permeability in bCLCA1 specific siRNA transfected cells. Baseline HCO(3)(-) permeability, however, was not different between controls and siRNA treated cells. We conclude that the calcium-activated chloride channel (bCLCA1) is expressed in bovine corneal endothelial cells and can contribute to Ca(2+) dependent apical HCO(3)(-) permeability, but not resting permeability, across the corneal endothelium.
Figures



Similar articles
-
Dependence of cAMP meditated increases in Cl- and HCO(3)- permeability on CFTR in bovine corneal endothelial cells.Exp Eye Res. 2008 Apr;86(4):684-90. doi: 10.1016/j.exer.2008.01.017. Epub 2008 Feb 2. Exp Eye Res. 2008. PMID: 18325495 Free PMC article.
-
Enhancement of HCO(3)(-) permeability across the apical membrane of bovine corneal endothelium by multiple signaling pathways.Invest Ophthalmol Vis Sci. 2002 Apr;43(4):1146-53. Invest Ophthalmol Vis Sci. 2002. PMID: 11923259
-
Expression, localization, and functional evaluation of CFTR in bovine corneal endothelial cells.Am J Physiol Cell Physiol. 2002 Apr;282(4):C673-83. doi: 10.1152/ajpcell.00384.2001. Am J Physiol Cell Physiol. 2002. PMID: 11880256 Free PMC article.
-
Identity and regulation of ion transport mechanisms in the corneal endothelium.Prog Retin Eye Res. 2003 Jan;22(1):69-94. doi: 10.1016/s1350-9462(02)00059-9. Prog Retin Eye Res. 2003. PMID: 12597924 Review.
-
Towards Clinical Application: Calcium Waves for In Vitro Qualitative Assessment of Propagated Primary Human Corneal Endothelial Cells.Cells. 2024 Dec 5;13(23):2012. doi: 10.3390/cells13232012. Cells. 2024. PMID: 39682760 Free PMC article. Review.
Cited by
-
Dependence of cAMP meditated increases in Cl- and HCO(3)- permeability on CFTR in bovine corneal endothelial cells.Exp Eye Res. 2008 Apr;86(4):684-90. doi: 10.1016/j.exer.2008.01.017. Epub 2008 Feb 2. Exp Eye Res. 2008. PMID: 18325495 Free PMC article.
-
Fluid transport by the cornea endothelium is dependent on buffering lactic acid efflux.Am J Physiol Cell Physiol. 2016 Jul 1;311(1):C116-26. doi: 10.1152/ajpcell.00095.2016. Epub 2016 May 25. Am J Physiol Cell Physiol. 2016. PMID: 27225657 Free PMC article.
-
Chloride channels and transporters in human corneal epithelium.Exp Eye Res. 2010 Jun;90(6):771-9. doi: 10.1016/j.exer.2010.03.013. Epub 2010 Mar 24. Exp Eye Res. 2010. PMID: 20346358 Free PMC article.
-
Role of carbonic anhydrase IV in corneal endothelial HCO3- transport.Invest Ophthalmol Vis Sci. 2008 Mar;49(3):1048-55. doi: 10.1167/iovs.07-1188. Invest Ophthalmol Vis Sci. 2008. PMID: 18326729 Free PMC article.
-
Molecular mechanisms underlying the corneal endothelial pump.Exp Eye Res. 2012 Feb;95(1):2-7. doi: 10.1016/j.exer.2011.06.004. Epub 2011 Jun 15. Exp Eye Res. 2012. PMID: 21693119 Free PMC article. Review.
References
-
- Bonanno JA. Identity and regulation of ion transport mechanisms in the corneal endothelium. Prog Retin Eye Res. 2003;22:69–94. - PubMed
-
- Bonanno JA, Giasson C. Intracellular pH regulation in fresh and cultured bovine corneal endothelium. II. Na:HCO3 cotransport and Cl/HCO3 exchange. Invest Ophthalmol Vis Sci. 1992;33:3068–3079. - PubMed
-
- Bonanno J, Guan Y, Jelamskii S, Kang X. Apical and basolateral CO2- permeability in cultured bovine corneal endothelial cells. Am J Physiol. 1999;277:C545–C553. - PubMed
-
- Bonanno JA, Yi G, Kang XJ, Srinivas SP. Reevaluation of exchange in cultured bovine corneal endothelial cells. Invest Ophthalmol Vis Sci. 1998;39:2713–2722. - PubMed
-
- Cunningham SA, Awayda MS, Bubien JK, Ismailov II, Arrate MP, Berdiev BK, Benos DJ, Fuller CM. Cloning of an epithelial chloride channel from bovine trachea. J Biol Chem. 1995;270:31016–31026. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

