Effective countermeasure against poisoning by organophosphorus insecticides and nerve agents
- PMID: 16914529
- PMCID: PMC1550772
- DOI: 10.1073/pnas.0605370103
Effective countermeasure against poisoning by organophosphorus insecticides and nerve agents
Abstract
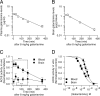
The nerve agents soman, sarin, VX, and tabun are deadly organophosphorus (OP) compounds chemically related to OP insecticides. Most of their acute toxicity results from the irreversible inhibition of acetylcholinesterase (AChE), the enzyme that inactivates the neurotransmitter acetylcholine. The limitations of available therapies against OP poisoning are well recognized, and more effective antidotes are needed. Here, we demonstrate that galantamine, a reversible and centrally acting AChE inhibitor approved for treatment of mild to moderate Alzheimer's disease, protects guinea pigs from the acute toxicity of lethal doses of the nerve agents soman and sarin, and of paraoxon, the active metabolite of the insecticide parathion. In combination with atropine, a single dose of galantamine administered before or soon after acute exposure to lethal doses of soman, sarin, or paraoxon effectively and safely counteracted their toxicity. Doses of galantamine needed to protect guinea pigs fully against the lethality of OPs were well tolerated. In preventing the lethality of nerve agents, galantamine was far more effective than pyridostigmine, a peripherally acting AChE inhibitor, and it was less toxic than huperzine, a centrally acting AChE inhibitor. Thus, a galantamine-based therapy emerges as an effective and safe countermeasure against OP poisoning.
Conflict of interest statement
Conflict of interest statement: No conflicts declared.
Figures




Comment in
-
Overcoming inhibitions.Proc Natl Acad Sci U S A. 2006 Sep 5;103(36):13263-4. doi: 10.1073/pnas.0606052103. Epub 2006 Aug 28. Proc Natl Acad Sci U S A. 2006. PMID: 16938861 Free PMC article. No abstract available.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

