A mycothiol synthase mutant of Mycobacterium tuberculosis has an altered thiol-disulfide content and limited tolerance to stress
- PMID: 16923891
- PMCID: PMC1595396
- DOI: 10.1128/JB.00393-06
A mycothiol synthase mutant of Mycobacterium tuberculosis has an altered thiol-disulfide content and limited tolerance to stress
Abstract
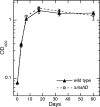
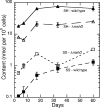
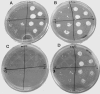
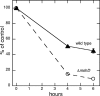
Mycothiol (MSH) (acetyl-Cys-GlcN-Ins) is the major low-molecular-mass thiol in Mycobacterium tuberculosis. MSH has antioxidant activity, can detoxify a variety of toxic compounds, and helps to maintain the reducing environment of the cell. The production of MSH provides a potential novel target for tuberculosis treatment. Biosynthesis of MSH requires at least four genes. To determine which of these genes is essential in M. tuberculosis, we have been constructing targeted gene disruptions. Disruption in the mshC gene is lethal to M. tuberculosis, while disruption in the mshB gene results in MSH levels 20 to 100% of those of the wild type. For this study, we have constructed a targeted gene disruption in the mshD gene that encodes mycothiol synthase, the final enzyme in MSH biosynthesis. The mshD mutant produced approximately 1% of normal MSH levels but high levels of the MshD substrate Cys-GlcN-Ins and the novel thiol N-formyl-Cys-GlcN-Ins. Although N-formyl-Cys-GlcN-Ins was maintained in a highly reduced state, Cys-GlcN-Ins was substantially oxidized. In both the wild type and the mshD mutant, cysteine was predominantly oxidized. The M. tuberculosis mshD mutant grew poorly on agar plates lacking catalase and oleic acid and in low-pH media and had heightened sensitivity to hydrogen peroxide. The inability of the mshD mutant to survive and grow in macrophages may be associated with its altered thiol-disulfide status. It appears that N-formyl-Cys-GlcN-Ins serves as a weak surrogate for MSH but is not sufficient to support normal growth of M. tuberculosis under stress conditions such as those found within the macrophage.
Figures

Similar articles
-
A mycothiol synthase mutant of Mycobacterium smegmatis produces novel thiols and has an altered thiol redox status.J Bacteriol. 2005 Nov;187(21):7309-16. doi: 10.1128/JB.187.21.7309-7316.2005. J Bacteriol. 2005. PMID: 16237013 Free PMC article.
-
Association of mycothiol with protection of Mycobacterium tuberculosis from toxic oxidants and antibiotics.Mol Microbiol. 2003 Mar;47(6):1723-32. doi: 10.1046/j.1365-2958.2003.03416.x. Mol Microbiol. 2003. PMID: 12622824
-
Mycothiol biochemistry.Arch Microbiol. 2002 Dec;178(6):388-94. doi: 10.1007/s00203-002-0469-4. Epub 2002 Sep 3. Arch Microbiol. 2002. PMID: 12420157 Review.
-
Identification of the mycothiol synthase gene (mshD) encoding the acetyltransferase producing mycothiol in actinomycetes.Arch Microbiol. 2002 Nov;178(5):331-7. doi: 10.1007/s00203-002-0462-y. Epub 2002 Aug 15. Arch Microbiol. 2002. PMID: 12375100
-
Biosynthesis and functions of mycothiol, the unique protective thiol of Actinobacteria.Microbiol Mol Biol Rev. 2008 Sep;72(3):471-94. doi: 10.1128/MMBR.00008-08. Microbiol Mol Biol Rev. 2008. PMID: 18772286 Free PMC article. Review.
Cited by
-
Mycobacterium tuberculosis Thioredoxin Reductase Is Essential for Thiol Redox Homeostasis but Plays a Minor Role in Antioxidant Defense.PLoS Pathog. 2016 Jun 1;12(6):e1005675. doi: 10.1371/journal.ppat.1005675. eCollection 2016 Jun. PLoS Pathog. 2016. PMID: 27249779 Free PMC article.
-
The Macrophage Response to Mycobacterium tuberculosis and Opportunities for Autophagy Inducing Nanomedicines for Tuberculosis Therapy.Front Cell Infect Microbiol. 2021 Feb 8;10:618414. doi: 10.3389/fcimb.2020.618414. eCollection 2020. Front Cell Infect Microbiol. 2021. PMID: 33628745 Free PMC article. Review.
-
Mycobacterium tuberculosis WhiB3 Responds to Vacuolar pH-induced Changes in Mycothiol Redox Potential to Modulate Phagosomal Maturation and Virulence.J Biol Chem. 2016 Feb 5;291(6):2888-903. doi: 10.1074/jbc.M115.684597. Epub 2015 Dec 4. J Biol Chem. 2016. PMID: 26637353 Free PMC article.
-
Divergent downstream biosynthetic pathways are supported by <sc>L</sc>-cysteine synthases of Mycobacterium tuberculosis.Elife. 2024 Aug 29;12:RP91970. doi: 10.7554/eLife.91970. Elife. 2024. PMID: 39207917 Free PMC article.
-
The role of thiols in antioxidant systems.Free Radic Biol Med. 2019 Aug 20;140:14-27. doi: 10.1016/j.freeradbiomed.2019.05.035. Epub 2019 Jun 13. Free Radic Biol Med. 2019. PMID: 31201851 Free PMC article. Review.
References
-
- Allen, B. W. 1998. Mycobacteria. General culture methodology and safety considerations. Methods Mol. Biol. 101:15-30. - PubMed
-
- Anderberg, S. J., G. L. Newton, and R. C. Fahey. 1998. Mycothiol biosynthesis and metabolism. Cellular levels of potential intermediates in the biosynthesis and degradation of mycothiol. J. Biol. Chem. 273:30391-30397. - PubMed
-
- Buchmeier, N., A. Blanc-Potard, S. Ehrt, D. Piddington, L. Riley, and E. A. Groisman. 2000. A parallel intraphagosomal survival strategy shared by Mycobacterium tuberculosis and Salmonella enterica. Mol. Microbiol. 35:1375-1382. - PubMed
-
- Buchmeier, N. A., G. L. Newton, T. Koledin, and R. C. Fahey. 2003. Association of mycothiol with protection of Mycobacterium tuberculosis from toxic oxidants and antibiotics. Mol. Microbiol. 47:1723-1732. - PubMed
-
- Fahey, R. C., and G. L. Newton. 1987. Determination of low-molecular-weight thiols using monobromobimane fluorescent labeling and high-performance liquid chromatography. Methods Enzymol. 143:85-96. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

