Residues involved in FecR binding are localized on one side of the FecA signaling domain in Escherichia coli
- PMID: 16923915
- PMCID: PMC1595354
- DOI: 10.1128/JB.00741-06
Residues involved in FecR binding are localized on one side of the FecA signaling domain in Escherichia coli
Abstract
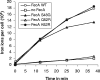
Ferric citrate transport in Escherichia coli involves proteins encoded by the fec genes, including the transport and signaling protein FecA and the signal transducing protein FecR. Randomly isolated FecA point mutants showed a reduced interaction with FecR and a reduced transcription initiation of the ferric citrate transport genes. The mutations were localized on one side of the FecA signaling domain, which might form the interface to FecR. Some of the mutants showed strongly reduced iron transport rates, which suggests that the signaling domain affects the structure of the FecA transporter domain.
Figures
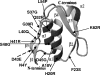
Similar articles
-
Occurrence and regulation of the ferric citrate transport system in Escherichia coli B, Klebsiella pneumoniae, Enterobacter aerogenes, and Photorhabdus luminescens.Arch Microbiol. 2005 Nov;184(3):175-86. doi: 10.1007/s00203-005-0035-y. Epub 2005 Nov 10. Arch Microbiol. 2005. PMID: 16193283
-
Sites of interaction between the FecA and FecR signal transduction proteins of ferric citrate transport in Escherichia coli K-12.J Bacteriol. 2003 Jul;185(13):3745-52. doi: 10.1128/JB.185.13.3745-3752.2003. J Bacteriol. 2003. PMID: 12813067 Free PMC article.
-
Control of the ferric citrate transport system of Escherichia coli: mutations in region 2.1 of the FecI extracytoplasmic-function sigma factor suppress mutations in the FecR transmembrane regulatory protein.J Bacteriol. 2001 Jan;183(1):162-70. doi: 10.1128/JB.183.1.162-170.2001. J Bacteriol. 2001. PMID: 11114913 Free PMC article.
-
Gene regulation by transmembrane signaling.Biometals. 2006 Apr;19(2):103-13. doi: 10.1007/s10534-005-8253-y. Biometals. 2006. PMID: 16718597 Review.
-
Surface signaling: novel transcription initiation mechanism starting from the cell surface.Arch Microbiol. 1997 Jun;167(6):325-31. doi: 10.1007/s002030050451. Arch Microbiol. 1997. PMID: 9148773 Review.
Cited by
-
Signal transduction pathway of TonB-dependent transporters.Proc Natl Acad Sci U S A. 2007 Jan 9;104(2):513-8. doi: 10.1073/pnas.0609887104. Epub 2006 Dec 29. Proc Natl Acad Sci U S A. 2007. PMID: 17197416 Free PMC article.
-
Signaling mechanisms for activation of extracytoplasmic function (ECF) sigma factors.Biochim Biophys Acta. 2008 Sep;1778(9):1930-45. doi: 10.1016/j.bbamem.2007.06.005. Epub 2007 Jun 15. Biochim Biophys Acta. 2008. PMID: 17673165 Free PMC article. Review.
-
Docking of the periplasmic FecB binding protein to the FecCD transmembrane proteins in the ferric citrate transport system of Escherichia coli.J Bacteriol. 2007 Oct;189(19):6913-8. doi: 10.1128/JB.00884-07. Epub 2007 Jul 27. J Bacteriol. 2007. PMID: 17660286 Free PMC article.
-
Transcription regulation of iron carrier transport genes by ECF sigma factors through signaling from the cell surface into the cytoplasm.FEMS Microbiol Rev. 2022 Jul 1;46(4):fuac010. doi: 10.1093/femsre/fuac010. FEMS Microbiol Rev. 2022. PMID: 35138377 Free PMC article.
-
New Insights into the Regulation of Cell-Surface Signaling Activity Acquired from a Mutagenesis Screen of the Pseudomonas putida IutY Sigma/Anti-Sigma Factor.Front Microbiol. 2017 May 2;8:747. doi: 10.3389/fmicb.2017.00747. eCollection 2017. Front Microbiol. 2017. PMID: 28512454 Free PMC article.
References
-
- Braun, V., and S. Mahren. 2005. Transmembrane transcriptional control (surface signalling) of the Escherichia coli Fec type. FEMS Microbiol. Rev. 29:673-684. - PubMed
-
- Dmitrova, M., G. Younès-Cauet, P. Oertel-Buchheit, D. Porte, M. Schnarr, and M. Granger-Schnarr. 1998. A new LexA-based genetic system for monitoring and analyzing protein heterodimerization in Escherichia coli. Mol. Gen. Genet. 257:205-212. - PubMed
-
- Ferguson, A., R. Chakraborty, B. Smith, L. Esser, D. van der Helm, and J. Deisenhofer. 2002. Structural basis of gating by the outer membrane transporter FecA. Science 295:1715-1719. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

