Cellular protein breakdown and systemic inflammation are unaffected by pulmonary rehabilitation in COPD
- PMID: 16928709
- PMCID: PMC2111241
- DOI: 10.1136/thx.2006.060368
Cellular protein breakdown and systemic inflammation are unaffected by pulmonary rehabilitation in COPD
Abstract
Background: Pulmonary rehabilitation can improve the functional capacity, but has a variable effect on the low fat-free mass (FFM) in patients with chronic obstructive pulmonary disease.
Hypothesis: Pulmonary rehabilitation would not affect catabolic drives such as systemic inflammation and also protein breakdown.
Methods: Patients (n = 40) were studied at the start of an 8-week in-patient pulmonary rehabilitation programme, at the end of the programme and 4 weeks later. FFM and functional capacity (quadriceps strength, handgrip strength and peak workload) were assessed. Pseudouridine (PSU) urinary excretion (cellular protein breakdown) and inflammatory status were determined. Healthy participants had a single baseline assessment (n = 18).
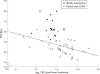
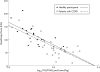
Results: PSU, (IL)-6 and soluble tumour necrosis factor (sTNF)alpha R75 were increased in patients compared with healthy participants, whereas FFM and functional capacity were reduced (all p < 0.01). PSU was inversely related to both FFM and skeletal muscle function. FFM and functional parameters increased with rehabilitation, but PSU and inflammatory status were unaffected. The gain in FFM was lost 4 weeks after the completion of rehabilitation (p < 0.01).
Conclusion: The anabolic effect of pulmonary rehabilitation improved FFM, but it did not reverse the increased protein breakdown or systemic inflammation. Thus, on cessation of pulmonary rehabilitation the FFM gains were lost owing to a loss of anabolic drive.
Conflict of interest statement
Competing interests: DJS has received research grants from AstraZeneca and GlaxoSmithKline. AMWJS has received research grants from GlaxoSmithkline and Numico Research. EFMW serves as a consultant to GlaxoSmithKline (GSK) and is a member of the scientific advisory board for GSK. He received lecture fees and research grants between 2001 and 2004 from GSK.
References
-
- Schols A M, Broekhuizen R, Weling‐Scheepers C A.et al Body composition and mortality in chronic obstructive pulmonary disease. Am J Clin Nutr 20058253–59. - PubMed
-
- Shoup R, Dalsky G, Warner S.et al Body composition and health‐related quality of life in patients with obstructive airways disease. Eur Respir J 1997101576–1580. - PubMed
-
- Bolton C E, Ionescu A A, Shiels K M.et al Associated loss of fat free mass and bone mineral density in chronic obstructive pulmonary disease. Am J Respir Crit Care Med 20041701286–1293. - PubMed
-
- Donahoe M, Rogers R M, Wilson D O.et al Oxygen consumption of the respiratory muscles in normal and in malnourished patients with chronic obstructive pulmonary disease. Am Rev Respir Dis 1989941260–1263. - PubMed
-
- Debigaré R, Marquis K, Côté C H.et al Catabolic/anabolic balance and muscle wasting in patients with COPD. Chest 200312483–89. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical