The affinity of the FimH fimbrial adhesin is receptor-driven and quasi-independent of Escherichia coli pathotypes
- PMID: 16930149
- PMCID: PMC1618777
- DOI: 10.1111/j.1365-2958.2006.05352.x
The affinity of the FimH fimbrial adhesin is receptor-driven and quasi-independent of Escherichia coli pathotypes
Abstract
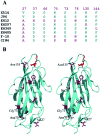
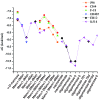
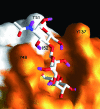
Type-1 fimbriae are important virulence factors for the establishment of Escherichia coli urinary tract infections. Bacterial adhesion to the high-mannosylated uroplakin Ia glycoprotein receptors of bladder epithelium is mediated by the FimH adhesin. Previous studies have attributed differences in mannose-sensitive adhesion phenotypes between faecal and uropathogenic E. coli to sequence variation in the FimH receptor-binding domain. We find that FimH variants from uropathogenic, faecal and enterohaemorrhagic isolates express the same specificities and affinities for high-mannose structures. The only exceptions are FimHs from O157 strains that carry a mutation (Asn135Lys) in the mannose-binding pocket that abolishes all binding. A high-mannose microarray shows that all substructures are bound by FimH and that the largest oligomannose is not necessarily the best binder. Affinity measurements demonstrate a strong preference towards oligomannosides exposing Manalpha1-3Man at their non-reducing end. Binding is further enhanced by the beta1-4-linkage to GlcNAc, where binding is 100-fold better than that of alpha-d-mannose. Manalpha1-3Manbeta1-4GlcNAc, a major oligosaccharide present in the urine of alpha-mannosidosis patients, thus constitutes a well-defined FimH epitope. Differences in affinities for high-mannose structures are at least 10-fold larger than differences in numbers of adherent bacteria between faecal and uropathogenic strains. Our results imply that the carbohydrate expression profile of targeted host tissues and of natural inhibitors in urine, such as Tamm-Horsfall protein, are stronger determinants of adhesion than FimH variation.
Figures

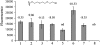
Similar articles
-
Receptor binding studies disclose a novel class of high-affinity inhibitors of the Escherichia coli FimH adhesin.Mol Microbiol. 2005 Jan;55(2):441-55. doi: 10.1111/j.1365-2958.2004.04415.x. Mol Microbiol. 2005. PMID: 15659162
-
Glycosylation changes as important factors for the susceptibility to urinary tract infection.Biochem Soc Trans. 2011 Jan;39(1):349-54. doi: 10.1042/BST0390349. Biochem Soc Trans. 2011. PMID: 21265802
-
Differential stability and trade-off effects of pathoadaptive mutations in the Escherichia coli FimH adhesin.Infect Immun. 2007 Jul;75(7):3548-55. doi: 10.1128/IAI.01963-06. Epub 2007 May 14. Infect Immun. 2007. PMID: 17502398 Free PMC article.
-
Molecular structure of adhesin domains in Escherichia coli fimbriae.Int J Med Microbiol. 2005 Oct;295(6-7):479-86. doi: 10.1016/j.ijmm.2005.06.010. Int J Med Microbiol. 2005. PMID: 16238022 Review.
-
Mannose-derived FimH antagonists: a promising anti-virulence therapeutic strategy for urinary tract infections and Crohn's disease.Expert Opin Ther Pat. 2016;26(2):175-97. doi: 10.1517/13543776.2016.1131266. Epub 2016 Jan 22. Expert Opin Ther Pat. 2016. PMID: 26651364 Review.
Cited by
-
A Novel Integrated Way for Deciphering the Glycan Code for the FimH Lectin.Molecules. 2018 Oct 28;23(11):2794. doi: 10.3390/molecules23112794. Molecules. 2018. PMID: 30373288 Free PMC article.
-
Glycomimetics for the inhibition and modulation of lectins.Chem Soc Rev. 2023 Jun 6;52(11):3663-3740. doi: 10.1039/d2cs00954d. Chem Soc Rev. 2023. PMID: 37232696 Free PMC article. Review.
-
Surfaces Presenting α-Phenyl Mannoside Derivatives Enable Formation of Stable, High Coverage, Non-pathogenic Escherichia coli Biofilms against Pathogen Colonization.Biomater Sci. 2015 Jun 1;3(6):781-880. doi: 10.1039/C5BM00076A. Biomater Sci. 2015. PMID: 26029359 Free PMC article.
-
Conserved FimH mutations in the global Escherichia coli ST131 multi-drug resistant lineage weaken interdomain interactions and alter adhesin function.Comput Struct Biotechnol J. 2022 Aug 20;20:4532-4541. doi: 10.1016/j.csbj.2022.08.040. eCollection 2022. Comput Struct Biotechnol J. 2022. PMID: 36090810 Free PMC article.
-
Adhesion of Enteropathogenic, Enterotoxigenic, and Commensal Escherichia coli to the Major Zymogen Granule Membrane Glycoprotein 2.Appl Environ Microbiol. 2022 Mar 8;88(5):e0227921. doi: 10.1128/aem.02279-21. Epub 2022 Jan 12. Appl Environ Microbiol. 2022. PMID: 35020452 Free PMC article.
References
-
- Abraham SN, Sun D, Dale JB, Beachey EH. Conservation of the d-mannose-adhesion protein among type 1 fimbriated members of the family Enterobactericeae. Nature. 1988;336:682–684. - PubMed
-
- Adams EW, Ratner DM, Bokesch HR, McMahon JB, O'Keefe BR, Seeberger PH. Oligosaccharide and glycoprotein microarrays as tools in HIV glycobiology: glycan-dependent gp120/protein interactions. Chem Biol. 2004;11:875–881. - PubMed
-
- Aronson M, Medalia O, Schori L, Mirelman D, Sharon N, Ofek I. Prevention of colonization of the urinary-tract by blocking bacterial adherence with methyl-α-d-mannopyranoside. Isr J Med Sci. 1979;15:88. - PubMed
-
- Bates JM, Raffi HM, Prasadan K, Mascarenhas R, Laszik Z, Maeda N, et al. Tamm-Horsfall protein knockout mice are more prone to urinary tract infection. Kidney Int. 2004;65:791–797. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

