Vascular remodeling is airway generation-specific in a primate model of chronic asthma
- PMID: 16931639
- PMCID: PMC2648107
- DOI: 10.1164/rccm.200506-848OC
Vascular remodeling is airway generation-specific in a primate model of chronic asthma
Abstract
Rationale: Changes in the density of bronchial vessels have been proposed as a part of airway remodeling that occurs in chronic asthma.
Objectives: Using an established nonhuman primate model of chronic allergic asthma, we evaluated changes in vascular density as well as the contribution of bronchial epithelium to produce vascular endothelial growth factor (VEGF).
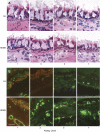
Methods: Eight juvenile rhesus macaques were divided into two groups of four. One group was exposed to 11 cycles of aerosolized house dust mite allergen (HDMA), whereas the other was exposed to filtered air. Bronchial wall vasculature was identified using an immunohistochemical approach, and vascular density was quantified stereologically. A semiquantitative polymerase chain reaction approach was used to estimate VEGF splice variant gene expression at discrete airway generations. Cell culture of primary tracheal epithelial cells with varying concentrations of HDMA was used to quantify the direct contribution of the epithelium to VEGF production.
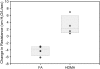
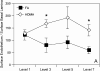
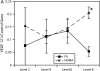
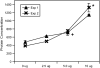
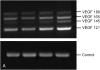
Results: Bronchial vascular density was increased at mid- to lower airway generations, which was independent of changes in the interstitial compartment. The VEGF121 splice variant was significantly increased at lower airway generations. VEGF protein increased in a dose-dependant fashion in vitro primarily by an increase in VEGF121 gene expression.
Conclusion: This study highlights that increased vascular density in an animal model of chronic allergic asthma is airway generation specific and associated with a unique increase of VEGF splice variant gene expression. Airway epithelium is the likely source for increased VEGF.
Figures






Similar articles
-
Gene expression of vascular endothelial growth factor and its receptors and angiogenesis in bronchial asthma.J Allergy Clin Immunol. 2001 Jun;107(6):1034-8. doi: 10.1067/mai.2001.115626. J Allergy Clin Immunol. 2001. PMID: 11398081
-
[The effect of a vascular endothelial growth factor receptor inhibitor on airway inflammation and airway remodeling in a murine model of asthma].Zhonghua Jie He He Hu Xi Za Zhi. 2005 Nov;28(11):755-9. Zhonghua Jie He He Hu Xi Za Zhi. 2005. PMID: 16324270 Chinese.
-
Vascular endothelial growth factor-mediated induction of angiogenesis by human rhinoviruses.J Allergy Clin Immunol. 2006 Feb;117(2):291-7. doi: 10.1016/j.jaci.2005.11.005. J Allergy Clin Immunol. 2006. PMID: 16461129
-
The non-human primate as a model for studying COPD and asthma.Pulm Pharmacol Ther. 2008 Oct;21(5):755-66. doi: 10.1016/j.pupt.2008.01.008. Epub 2008 Feb 1. Pulm Pharmacol Ther. 2008. PMID: 18339566 Review.
-
Angiogenesis and lymphangiogenesis in bronchial asthma.Allergy. 2010 Aug;65(8):946-58. doi: 10.1111/j.1398-9995.2010.02372.x. Epub 2010 Apr 23. Allergy. 2010. PMID: 20415716 Review.
Cited by
-
Lung effects of inhaled corticosteroids in a rhesus monkey model of childhood asthma.Clin Exp Allergy. 2012 Jul;42(7):1104-18. doi: 10.1111/j.1365-2222.2012.04005.x. Clin Exp Allergy. 2012. PMID: 22702509 Free PMC article.
-
Proteolytic Activity Present in House-Dust-Mite Extracts Degrades ENA-78/CXCL5 and Reduces Neutrophil Migration.J Allergy (Cairo). 2014;2014:673673. doi: 10.1155/2014/673673. Epub 2014 May 4. J Allergy (Cairo). 2014. PMID: 24883064 Free PMC article.
-
Interpretation of Clinical Efficacy of Biologics in Chronic Rhinosinusitis With Nasal Polyps via Understanding the Local and Systemic Pathomechanisms.Allergy Asthma Immunol Res. 2022 Sep;14(5):465-478. doi: 10.4168/aair.2022.14.5.465. Allergy Asthma Immunol Res. 2022. PMID: 36174990 Free PMC article. Review.
-
SMAD Signaling in the Airways of Healthy Rhesus Macaques versus Rhesus Macaques with Asthma Highlights a Relationship Between Inflammation and Bone Morphogenetic Proteins.Am J Respir Cell Mol Biol. 2016 Apr;54(4):562-73. doi: 10.1165/rcmb.2015-0210OC. Am J Respir Cell Mol Biol. 2016. PMID: 26414797 Free PMC article.
-
Understanding the mechanisms of viral induced asthma: new therapeutic directions.Pharmacol Ther. 2008 Mar;117(3):313-53. doi: 10.1016/j.pharmthera.2007.11.002. Epub 2008 Jan 29. Pharmacol Ther. 2008. PMID: 18234348 Free PMC article. Review.
References
-
- Hoshino M, Nakamura Y, Hamid QA. Gene expression of vascular endothelial growth factor and its receptors and angiogenesis in bronchial asthma. J Allergy Clin Immunol 2001;107:1034–1038. - PubMed
-
- McLaughlin RF, Tyler WS, Canada RO. A study of the subgross pulmonary anatomy in various mammals. Am J Anat 1961;108:149–165.
-
- Tischer E, Mitchell R, Hartman T, Silva M, Gospodarowicz D, Fiddes JC, Abraham JA. The human gene for vascular endothelial growth factor: multiple protein forms are encoded through alternative exon splicing. J Biol Chem 1991;266:11947–11954. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

