A randomized, multicenter study to determine the safety and efficacy of the immunoconjugate SGN-15 plus docetaxel for the treatment of non-small cell lung carcinoma
- PMID: 16934909
- PMCID: PMC3715069
- DOI: 10.1016/j.lungcan.2006.05.020
A randomized, multicenter study to determine the safety and efficacy of the immunoconjugate SGN-15 plus docetaxel for the treatment of non-small cell lung carcinoma
Abstract
Purpose: Chemotherapy prolongs survival and improves quality of life (QOL) for good performance status (PS) patients with advanced non-small cell lung cancer (NSCLC). Targeted therapies may improve chemotherapy effectiveness without worsening toxicity. SGN-15 is an antibody-drug conjugate (ADC), consisting of a chimeric murine monoclonal antibody recognizing the Lewis Y (Le(y)) antigen, conjugated to doxorubicin. Le(y) is an attractive target since it is expressed by most NSCLC. SGN-15 was active against Le(y)-positive tumors in early phase clinical trials and was synergistic with docetaxel in preclinical experiments. This Phase II, open-label study was conducted to confirm the activity of SGN-15 plus docetaxel in previously treated NSCLC patients.
Experimental design: Sixty-two patients with recurrent or metastatic NSCLC expressing Le(y), one or two prior chemotherapy regimens, and PS< or =2 were randomized 2:1 to receive SGN-15 200 mg/m2/week with docetaxel 35 mg/m2/week (Arm A) or docetaxel 35 mg/m2/week alone (Arm B) for 6 of 8 weeks. Intrapatient dose-escalation of SGN-15 to 350 mg/m2 was permitted in the second half of the study. Endpoints were survival, safety, efficacy, and quality of life.
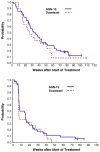
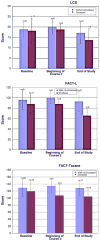
Results: Forty patients on Arm A and 19 on Arm B received at least one treatment. Patients on Arms A and B had median survivals of 31.4 and 25.3 weeks, 12-month survivals of 29% and 24%, and 18-month survivals of 18% and 8%, respectively. Toxicity was mild in both arms. QOL analyses favored Arm A.
Conclusions: SGN-15 plus docetaxel is a well-tolerated and active second and third line treatment for NSCLC patients. Ongoing studies are exploring alternate schedules to maximize synergy between these agents.
Conflict of interest statement
All authors, with the exception of those employed by Seattle Genetics, have indicated no potential conflicts of interest. The employees of or contractors to Seattle Genetics include Laurie Grove, Michael Thorn, Dennis Miller, and Jonathan Drachman.
Figures
Similar articles
-
Phase II randomized trial of tri-weekly versus days 1 and 8 weekly docetaxel as a second-line treatment of advanced non-small cell lung cancer.Jpn J Clin Oncol. 2005 Dec;35(12):700-6. doi: 10.1093/jjco/hyi191. Epub 2005 Nov 22. Jpn J Clin Oncol. 2005. PMID: 16303792 Clinical Trial.
-
Randomised phase II trial of 4 dose levels of single agent docetaxel in performance status (PS) 2 patients with advanced non-small cell lung cancer (NSCLC): DOC PS2 trial. Manchester lung cancer group.Lung Cancer. 2011 Sep;73(3):338-44. doi: 10.1016/j.lungcan.2011.01.001. Epub 2011 Feb 5. Lung Cancer. 2011. PMID: 21296449 Clinical Trial.
-
Weekly docetaxel vs. docetaxel-based combination chemotherapy as second-line treatment of advanced non-small-cell lung cancer patients. The DISTAL-2 randomized trial.Lung Cancer. 2009 Feb;63(2):251-8. doi: 10.1016/j.lungcan.2008.05.027. Epub 2008 Jul 15. Lung Cancer. 2009. PMID: 18632181 Clinical Trial.
-
Docetaxel for previously treated non-small-cell lung cancer.Oncology (Williston Park). 2002 Jun;16(6 Suppl 6):45-51. Oncology (Williston Park). 2002. PMID: 12108897 Review.
-
Docetaxel in the treatment of advanced non-small-cell lung cancer.Expert Rev Anticancer Ther. 2008 Aug;8(8):1207-22. doi: 10.1586/14737140.8.8.1207. Expert Rev Anticancer Ther. 2008. PMID: 18699760 Review.
Cited by
-
A population-based clinical trial of irinotecan and Carboplatin.J Oncol. 2009;2009:458528. doi: 10.1155/2009/458528. Epub 2009 Apr 23. J Oncol. 2009. PMID: 19404408 Free PMC article.
-
Targeting Tumor Glycans for Cancer Therapy: Successes, Limitations, and Perspectives.Cancers (Basel). 2022 Jan 27;14(3):645. doi: 10.3390/cancers14030645. Cancers (Basel). 2022. PMID: 35158915 Free PMC article. Review.
-
Exploration of the antibody-drug conjugate clinical landscape.MAbs. 2023 Jan-Dec;15(1):2229101. doi: 10.1080/19420862.2023.2229101. MAbs. 2023. PMID: 37639687 Free PMC article. Review.
-
[DNA Damage Repair System and Antineoplastic Agents in Lung Cancer].Zhongguo Fei Ai Za Zhi. 2022 Jun 20;25(6):434-442. doi: 10.3779/j.issn.1009-3419.2022.101.24. Zhongguo Fei Ai Za Zhi. 2022. PMID: 35747923 Free PMC article. Review. Chinese.
-
Tumor-Associated Glycans as Targets for Immunotherapy: The Wistar Institute Experience/Legacy.Monoclon Antib Immunodiagn Immunother. 2021 Jun;40(3):89-100. doi: 10.1089/mab.2021.0024. Monoclon Antib Immunodiagn Immunother. 2021. PMID: 34161162 Free PMC article. Review.
References
-
- Cancer facts and figures. American Cancer Society; 2004.
-
- Landis SH, Murray T, Bolden S, Wingo PA. Cancer statistics 1999. CA Cancer J Clin. 1999;49:8–31. 1. - PubMed
-
- Ginsberg RJ. Continuing controversies in staging NSCLC: an analysis of the revised 1997 staging system. Oncology (Williston Park) 1998;12:51–4. - PubMed
-
- Gandara DR, Chansky K, Albain KS, et al. Consolidation docetaxel after concurrent chemoradiotherapy in stage IIIB non-small-cell lung cancer: phase II Southwest Oncology Group Study S9504. J Clin Oncol. 2003;21:2004–10. - PubMed
-
- Schiller JH, Harrington D, Belani CP, et al. Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer. N Engl J Med. 2002;346:92–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

