Threonine-rich repeats increase fibronectin binding in the Candida albicans adhesin Als5p
- PMID: 16936142
- PMCID: PMC1595330
- DOI: 10.1128/EC.00120-06
Threonine-rich repeats increase fibronectin binding in the Candida albicans adhesin Als5p
Abstract
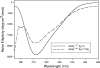
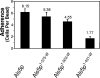
Commensal and pathogenic states of Candida albicans depend on cell surface-expressed adhesins, including those of the Als family. Mature Als proteins consist of a 300-residue N-terminal region predicted to have an immunoglobulin (Ig)-like fold, a 104-residue conserved Thr-rich region (T), a central domain of a variable number of tandem repeats (TR) of a 36-residue Thr-rich sequence, and a heavily glycosylated C-terminal Ser/Thr-rich stalk region, also of variable length (N. K. Gaur and S. A. Klotz, Infect. Immun. 65: 5289-5294, 1997). Domain deletions in ALS5 were expressed in Saccharomyces cerevisiae to excrete soluble protein and for surface display. Far UV circular dichroism indicated that soluble Ig-T showed a single negative peak at 212 nm, consistent with previous data indicating that this region has high beta-sheet content with very little alpha-helix. A truncation of Als5p with six tandem repeats (Ig-T-TR(6)) gave spectra with additional negative ellipticity at 200 nm and, at 227 to 240 nm, spectra characteristic of a structure with a similar fraction of beta-sheet but with additional structural elements as well. Soluble Als5p Ig-T and Ig-T-TR(6) fragments bound to fibronectin in vitro, but the inclusion of the TR region substantially increased affinity. Cellular adhesion assays with S. cerevisiae showed that the Ig-T domain mediated adherence to fibronectin and that TR repeats greatly increased cell-to-cell aggregation. Thus, the TR region of Als5p modulated the structure of the Ig-T region, augmented cell adhesion activity through increased binding to mammalian ligands, and simultaneously promoted fungal cell-cell interactions.
Figures






References
-
- Calderone, R. A. 2002. Candida and candidiasis. ASM Press, Washington, D.C.
-
- Chen, M. H., Z. M. Shen, S. Bobin, P. C. Kahn, and P. N. Lipke. 1995. Structure of Saccharomyces cerevisiae alpha-agglutinin. Evidence for a yeast cell wall protein with multiple immunoglobulin-like domains with atypical disulfides. J. Biol. Chem. 270:26168-26177. - PubMed
-
- Chou, P. Y., and G. D. Fasman. 1978. Prediction of the secondary structure of proteins from their amino acid sequence. Adv. Enzymol. Relat. Areas Mol. Biol. 47:45-148. - PubMed
-
- Dubois, M., K. Gilles, J. K. Hamilton, P. A. Rebers, and F. Smith. 1951. A colorimetric method for the determination of sugars. Nature 168:167. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

